The CBC model shows that within the chemistry of Mn, 79% of compounds fall into the
Question:
The CBC model shows that within the chemistry of Mn, ≈ 79% of compounds fall into the MnL5X class. Show that each of the following fits this description and that each has a value of EN = 18.
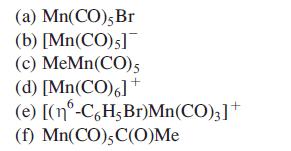
Transcribed Image Text:
(a) Mn(CO), Br (b) [Mn(CO) s] (c) MeMn(CO) 5 (d) [Mn(CO),]+ (e) [(nº-C6H, Br)Mn(CO)3] + (f) Mn(CO), C(O)Me
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (9 reviews)
To determine whether each of the given compounds fits into the MnL5X class and has a value of EN 18 we need to understand what the MnL5X class represe...View the full answer

Answered By

User l_1006857
I am a computer science professional with expertise in databases, AI programming, data structures and algorithms, and mathematics. With a strong background in these areas, I possess the knowledge and skills necessary to design and optimize database systems, develop intelligent algorithms and models, and solve complex computational problems. My proficiency in SQL, NoSQL, machine learning techniques, and mathematical concepts equips me to contribute to innovative projects and drive technological advancements.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
List three specific parts of the Case Guide, Objectives and Strategy Section (See below) that you had the most difficulty understanding. Describe your current understanding of these parts. Provide...
-
Planning is one of the most important management functions in any business. A front office managers first step in planning should involve determine the departments goals. Planning also includes...
-
The Crazy Eddie fraud may appear smaller and gentler than the massive billion-dollar frauds exposed in recent times, such as Bernie Madoffs Ponzi scheme, frauds in the subprime mortgage market, the...
-
Now apply one of those decision making models (Philosophical) to this scenario: Scenario: You are a CPA in the accounting department at a HealthCare System with three other accountants. You are good...
-
Condensed financial data of Turner Inc. follow. Additional information: 1. New plant assets costing $90,000 were purchased for cash during the year. 2. Old plant assets having an original cost of...
-
In problem, find an equation of the line L. L is perpendicular to y = 2x (1, 2) -1 - 2xF 3
-
Nereus Montemayor was an employee of VZ Hogs, a company that raises hogs and produces hog feed. VZ Hogs used an extruder manufactured by Sebright Products, Inc. to create hog feed out of discarded...
-
Presented below is information related to Dickinson Company for 2012. Retained earnings balance, January 1, 2012 $ 980,000 Sales revenue 25,000,000 Cost of goods sold 16,000,000 Interest revenue...
-
Explain the relationship and the difference between online analytical processing systems and customer relationship management systems within a business intelligence program.?
-
What is a synergic effect, and how does it relate to metalcarbonyl bonding?
-
Comment on: (a) The use of trimethylorganometallics in the manufacture of IIIV semiconductors; (b) The application of R 3 Al compounds as catalysts.
-
Do the following for each of the four data sets: a. Create a scatterplot of y versus x with a regression line ( = b0 + b1x), a plot of residuals versus the explanatory variable, a plot of residuals...
-
What is the area between the graphs of f(x) = 3x +5, g(x) = 2x 3, x = 2, and x = 12? -
-
Critically evaluate the important elements that make a group or team function effectively in the context of work and the organisation.
-
1) State the type number of the system. b. Determine the values for and 0 for this second-order system. c. Determine the percent overshoot for a step-function input to the system. d. Sketch the...
-
Evaluate the permutation. P(38,18) P(38,18) = (Use scientific notation. Use the multiplication syr
-
Develop a financial plan for implementation of new technology for a specific operating department of the hotel.
-
In 2013, Fred invested $50,000 in a general partnership. Freds interest is not considered to be a passive activity. If his share of the partnership losses is $35,000 in 2013 and $25,000 in 2014, how...
-
Annual dividends of ATTA Corp grew from $0.96 in 2005 to $1.76 in 2017. What was the annual growth rate?
-
Draw the carbocation intermediate generated by each of the following substrates in an E1 reaction: a. b. c. d. Br CI
-
Draw the carbocation intermediate generated when each of the following substrates is treated with sulfuric acid: a.
-
Identify the major and minor products for each of the following E1 reactions: a. b. c. d. H,SO. eat ETOH Heat Br
-
Gamma is a company based in Poland. The company expects to receive 1,500,000 in six months from a foreign customer. The company is concerned that euro may depreciate against Polish zloty before the...
-
The investor position in options for 100,000 units of the underlying instrument has the following characteristics: the delta is -30000 and the gamma is -80000. The current price of the underlying...
-
Compute the ERP for SBUX. Use the store data to calculate risk exposure. Stores ERP U.S. 8222 4.40% Canada 1083 4.40% Brazil 108 8.60% China 1540 5.30% Japan 1218 5.40% Thailand 312 6.80% U.K. 345...

Study smarter with the SolutionInn App


