Use data in Tables 6.5 and 6.6 to estimate the lattice energies of crystalline (a) [NH 4
Question:
Use data in Tables 6.5 and 6.6 to estimate the lattice energies of crystalline
(a) [NH4][PF6],
(b) K2[Cr2O7]
(c) BaS.
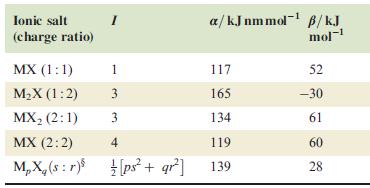
Table 6.5
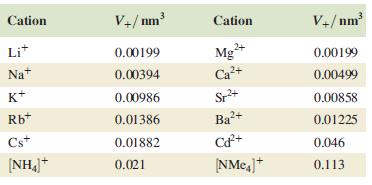
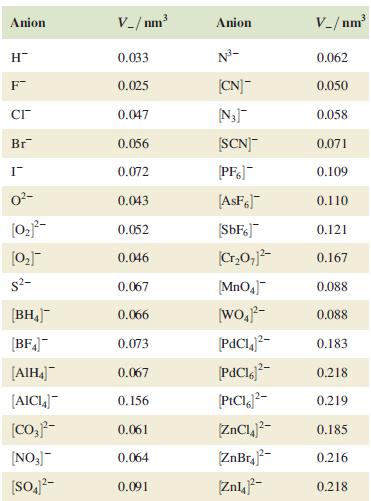
Table 6.6.
Transcribed Image Text:
Cation Lit Nat K+ Rb+ Cst [NH₁]+ V+/nm³ 0.00199 0.00394 0.00986 0.01386 0.01882 0.021 Cation Mg²+ Sr²+ Ba²+ Cd²+ |NMea]* V+/nm³ 0.00199 0.00499 0.00858 0.01225 0.046 0.113
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 55% (9 reviews)
To estimate the lattice energies of the given compounds using the data in Tables 65 and 66 we can use the BornHaber cycle ...View the full answer

Answered By

Abdul Wahab Qaiser
Before working at Mariakani, I volunteered at a local community center, where I tutored students from diverse backgrounds. I helped them improve their academic performance and develop self-esteem and confidence. I used creative teaching methods, such as role-playing and group discussions, to make the learning experience more engaging and enjoyable.
In addition, I have conducted workshops and training sessions for educators and mental health professionals on various topics related to counseling and psychology. I have presented research papers at conferences and published articles in academic journals.
Overall, I am passionate about sharing my knowledge and helping others achieve their goals. I believe that tutoring is an excellent way to make a positive impact on people's lives, and I am committed to providing high-quality, personalized instruction to my students.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The lattice energies of KBr and CsCl are nearly equal (Table 8.2).What can you conclude from this observation? TABLE 8.2 Lattice Energies for Some Ionic Compounds Lattice Energy Lattice Energy...
-
Use data in Tables 25.1 and 25.2, and Eq. (25.12) to construct the local volatility surface. In order to do it, set upa uniform grid, then use bi-cubic sp line interpolation/extrapolation to find the...
-
Cryptands such as crypt-[222] (see Fig. 11.9) are encapsulating ligands for s-block metal ions. Use the VBT approach to estimate values for the lattice energies of KMnO 4 and [K(crypt-[222])][MnO 4...
-
A strain relaxation test and a creep test are being conducted on a mouse Achilles tendon. If a strain of \epsi 0 = 60% is applied in the strain relaxation test and a stress of \ sigma 0 = 1 MPa is...
-
1. Why has there been a corporate governance "revolution" throughout the world recently? 2. What are the main areas of deficiencies as pointed out by corporate governance reformers? 3. Representing...
-
A partial (5 subterminal) nucleotide sequence of a prokaryotic mRNA is as follows: 5-.....AGGAGGCUCGAACAUGUCAAUAUGCUUGUUCCAAUCGUUAGCUGCGCAGGACCGUCCCGGA.. . . . . 3 When this mRNA is translated, what...
-
Fraud deterrence is centered on the fear of getting caught and the fear of getting punished. In your opinion, which is stronger and why?
-
Electro Repairs & Service, an electronics repair store, prepared the unadjusted trial balance shown below at the end of its first year of operations. For preparing the adjusting entries, the...
-
Consider again the market studied in Question 2, The demand curve is given by P = 100 Q and the marginal cost curve is given by MC = 10. This time we are going to assume that firms must invest funds...
-
Data from Tables 6.3 and 6.4 are needed for this problem. (a) Estimate the lattice energy of CsCl if the CsCl internuclear distance is 356.6 pm. (b) Now consider a polymorph of CsCl that crystallizes...
-
(a) Given that U(0 K) and f H(298 K) for MgO are 3795 and 602 kJ mol 1 respectively, derive a value for EA H(298 K) for the reaction: Other data: see Appendices. (b) Compare the calculated value...
-
If possible, solve the system. X y 2x + y I 2=-3 = -2 =-5
-
What, in words, is the fundamental relationship (equation) used in making percent of sales forecasts?
-
What is the main difference between an investment banker and a commercial banker?
-
Over long periods of time, is the real rate of return higher on 30-year Treasury bonds or 30-year Aaa corporate bonds?
-
What is meant by the term private placement? What are its advantages and disadvantages within the financial markets?
-
Distinguish between the concepts of the maturity-risk premium and the liquidity-risk premium.
-
Explain when the units-of-production method of depreciation would be appropriate to use and what kind of units a company could consider using.
-
Select a mass spectrometric technique with the highest mass resolution for identifying an unknown compound being eluted from a liquid chromatography column
-
Draw the B 12 unit that is a common motif of boron structures; take a viewpoint along a C 2 axis.
-
Give balanced chemical equations for the synthesis of 1,2-B 10 C 2 H 10 (Si(CH 3 ) 3 ) 2 starting with decaborane(14) and other reagents of your choice.
-
Give the IUPAC names of (a) B 10 H 14 , (b) [B 12 H 12 ] 2 , (c) Arachno-[B 12 H 14 ] 2 .
-
The financing provided for start-up, often high-risk, private business enterprises is called: Question 3 options: Venture capital. Junk bonds. Flotation costs. Initial public offerings. Financial...
-
What if you decide to hold the stock for two years? - D = $2.00 CF - D = $2.10 - P = $14.70 - = $2.00 } CF2 = $2.10 + $14.70 = $16.80 Now how much would you be willing to pay?
-
Shamrock Inc. was supposed to have received a payment of $25,000, 5 years ago, and $14,000, 2 years ago, from a customer who could not make either payment as scheduled. If the customer would like to...

Study smarter with the SolutionInn App


