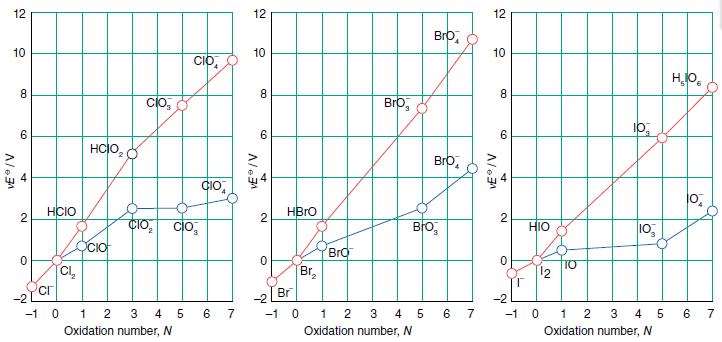
Use either the Frost diagram in Figure 17.14 or the Latimer diagrams in Resource section 3 to
Question:
Use either the Frost diagram in Figure 17.14 or the Latimer diagrams in Resource section 3 to calculate the standard potentials for the following couples in basic solution:
(a) ClO4−/ ClO− ,
(b) BrO4−/BrO− ,
(c) IO4− / IO−.
Comment on the relative feasibilities of the reduction reactions.
Figure 17.14.
Transcribed Image Text:
12 10 VEⓇ/V 8 6 2 0 HCIO СГ -1 0 2 Cl₂ HCIO, OCIO CIO CIO CIO₂ clo, CIO 1 2 3 4 5 6 7 Oxidation number, N 12 10 VEⓇ/V 8 6 4 2 0 HBrO Br -1 0 N õ Bro Bro Bro Bro BIO, 1 2 3 4 5 6 7 Oxidation number, N 12 10 VEⓇ/V 8 6 4 2 0 N -1 HIO 12 10 10₂ H₂O 10% 0 1 2 3 4 5 6 7 Oxidation number, N
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (4 reviews)
To calculate the standard potentials for the given couples in basic solution pH 7 we can use the Latimer diagrams The standard potentials E for halfre...View the full answer

Answered By

User l_998468
I have extensive tutoring experience, having worked as a private tutor for over three years. I have tutored students from different academic levels, including high school, undergraduate, and graduate levels. My tutoring experience has taught me to be patient, attentive to student needs, and effective in communicating difficult concepts in simple terms.
I have a strong background in statistics, probability theory, data analysis, and data visualization. I am proficient in using statistical software such as R, Python, and SPSS, which are commonly used in academic research and data analysis. Additionally, I have excellent communication and interpersonal skills, which enable me to establish rapport with students, understand their learning styles, and adapt my teaching approach to meet their needs.
I am passionate about teaching and helping students achieve their academic goals.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Inorganic Chemistry
ISBN: 9780198768128
7th Edition
Authors: Mark Weller, Tina Overton, Jonathan Rourke
Question Posted:
Students also viewed these Sciences questions
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
Answer the following questions using the Frost diagram in Fig. 6.20. (a) What are the consequences of dissolving Cl 2 in aqueous basic solution? (b) What are the consequences of dissolving Cl 2 in...
-
The following Latimer diagrams show the standard reduction potentials E /V for some oxidation states of iron in acid and alkaline solution: (a) Plot a Frost diagram showing the states of Fe under...
-
Using the DJIA data in Problem 5-39, use exponential smooth with trend adjustment to forecast the opening DJIA value for 2014. Use α = 0.8 and β = 0.2. Compare the MSE for...
-
On a balance sheet, what valuation must be reported for short-term investments in trading securities?
-
Timetrics 2016 survey of insurance professionals explores the use of technology in the industry. The file Technologies contains the responses to the question that asked what technologies these...
-
Steven Sanchez worked as a warehouse employee for Gruma Corporation. In December 2016, Sanchez was presented an arbitration agreement during a meeting with George Tate, the human resources manager...
-
Georgia Temps, a large labor contractor, supplies contract labor to building-construction companies. For 2014, Georgia Temps has budgeted to supply 84,000 hours of contract labor. Its variable costs...
-
Date Account Title Debit Credit Sept 1 Cash 10,000 Common Stock 10,000 Sept 2 Equipment 5,000 Cash 5,000 Sept 3 Rent Expense 400 Cash 400 Sept 4 Supplies 500 Cash 500 Sept 5 Advertising Expense 250...
-
Sketch all the isomers of the complexes [CrCl 4 F 2 ] 3 and [CrCl 3 F 3 ] 3 . Indicate how many fluorine environments would be indicated in the 19 F-NMR spectrum of each isomer.
-
Indicate the product of the reaction between ClF 5 and SbF 5 . Predict the shapes of the reactants and products.
-
At the date of a short sale, Sylvia has held substantially identical securities for more than 12 months. What is the nature of any gain or loss from the close of her short sale?
-
TTC Corporation purchased equipment on January 1 at a cost of $75,000. When purchased, the equigment years and a residual value of $15,000. Assuming TTC utilized the straight-line method of...
-
A large annuity company holds many industry group stocks. Among the industries are banks, business services and construction. Seven companies from each industry group are randomly sampled to test the...
-
When the Green Party of Alberta decides that it will allocate money in their next budget to infrastructure, this means that those resources are no longer available for spending on education or...
-
1. CONSIDER NARRATION: How would the story change if Faulkner told the story himself, in his own voice, rather than through a narrator who is one of the townspeople? 2. CONSIDER SYMBOLISM: How is...
-
Is there an argument in BMI(1979 link: https://supreme.justia.com/cases/federal/us/441/1/) that the blanket license raises output and that price-fixing is required for blanket license? Output of what...
-
Flight Operations The table below lists the times (min) required for randomly selected flights to taxi out for takeoff and the corresponding times (min) required to taxi in after landing. (See Data...
-
The executor of Gina Purcells estate has recorded the following information: Assets discovered at death (at fair value): Cash . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . ....
-
Lead-acid batteries accounted for 69% of all lead consumed in the US in 2015. (a) Complete the cell reaction given below (not balanced on the left-hand side) and show that the oxidation state changes...
-
Using bond enthalpy terms from Tables 14.2 and 15.3, estimate values of r H for the following reactions (a) 2N 2 N 4 (tetrahedral structure); (b) 2P 2 P 4 (tetrahedral structure); (c) 2C 2 H 2 C...
-
The glass industry manufactures millions of tonnes of glass per year. (a) Only certain element oxides form glasses. Explain why this is, giving examples of what are termed in the glass industry as...
-
prepare a two page paper on how these three principles apply in the supervision of others. Is one more important than the others? Define each term and analyze how each one effects the supervisory...
-
Activity-Based Product Costing Mello Manufacturing Company is a diversified manufacturer that manufactures three products (Alpha, Beta, and Omega) in a continuous production process. Senior...
-
Make your own Business Cycle! 1. Get a piece of paper 2. Draw a business cycle on your paper (large enough so there is part of the cycle in each box) 3. In each box i. Name and define that part of...

Study smarter with the SolutionInn App


