Use the Latimer diagrams to determine which species of N and P disproportionate in acid conditions. Acidic
Question:
Use the Latimer diagrams to determine which species of N and P disproportionate in acid conditions.
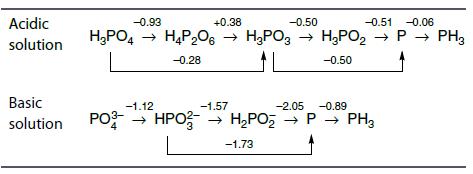
Transcribed Image Text:
Acidic solution -0.93 +0.38 -0.50 -0.51 -0.06 H₂PO4 → H4P₂06 → H3PO3 → H₂PO₂ → P→ PH3 -0.28 -0.50 Basic -1.57 -2.05 -0.89 solution PO3- HPO → H₂PO₂ → H₂PO₂ → P→ PH3 -1.73 -1.12
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 33% (3 reviews)
In the Latimer diagrams provided for nitrogen N and phosphorus P species the species that disproport...View the full answer

Answered By

Deepak Sharma
0.00
0 Reviews
10+ Question Solved
Related Book For 

Inorganic Chemistry
ISBN: 9780198768128
7th Edition
Authors: Mark Weller, Tina Overton, Jonathan Rourke
Question Posted:
Students also viewed these Sciences questions
-
Figure is a digraph representing a food web in a small ecosystem. A directed edge from a to b indicates that a has b as a source of food. Construct the adjacency matrix A for this digraph and use it...
-
Find the mean, variance, and standard deviation for each of the values of n and p when the conditions for the binomial distribution are met. a. n = 100, p = 0.75 b. n = 300, p = 0.3 c. n = 20, p =...
-
Find the mean, variance, and standard deviation for each of the values of n and p when the conditions for the binomial distributions are met. a. n = 1000, p = 0.1 b. n = 500, p = 0.25 c. n = 50, p =...
-
Suppose that the vice president of marketing asks you to write a program to create labels for a onetime advertising promotion. As IT manager, you know that the labels can be prepared more efficiently...
-
Indio Palms College wishes to monitor the efficiency and quality of its course registration process. a. List three input and three output measures for this process. b. Why would Indio Palms College...
-
Identify what items are treated as prior period adjustments.
-
A company wishes to hedge its exposure to a new fuel whose price changes have a 0.6 correlation with gasoline futures price changes. The company will lose $1 million for each 1 cent increase in the...
-
Leach Inc. experienced the following events for the first two years of its operations: 2016: 1. Issued $10,000 of common stock for cash. 2. Provided $78,000 of services on account. 3. Provided...
-
Solve the following DES. a) y (4) - (3) -3y"+5y'-2y=0
-
Write the balanced chemical equation corresponding to the standard enthalpy of formation of P 4 O 10 (s). Specify the structure, physical state (s, l, or g), and allotrope of the reactants. Does...
-
Starting with NH 3 (g) and other reagents of your choice, give the chemical equations and conditions for the synthesis of (a) HNO 3 , (b) NO 2 , (c) NH 2 OH, (d) N 3 .
-
Name the metric unit you would use to measure each of the quantities in Problems 5-10. a. The weight of a pencil gram b. The weight of an automobile kilogram
-
(WAN) T1 circuit transmits data via a frame of 193 bit shown as the below figure. (1) Please calculate the T1's data rate? (2) Please calculate the data rate of transmitting the Frame Bit S? Frame...
-
On July 31, 2021, Hummbug Corporation issued $100,000, 5%, 20-year bonds for $113,678, when the market interest rate was 4%. The bonds pay semi-annual interest on July 31 and January 31. Hummbug uses...
-
For a gauge pressure at A of -10.89 kPa, find the specific gravity of the gauge liquid L in the figure below. air 3.429m 3.2m +A 3.048m 2.743m Liquid L S.G.=1.6
-
Jesus, a soft-spoken employee of DCS Sanitation Management, Inc., the company that IBP uses in many of its plants, told me that every night on the job he gets terrible headaches. "You feel it in your...
-
The process varies depending on the group being surveyed, such as family members, communities, or select groups involved in specific markets like insurance or healthcare. They also use existing...
-
The accompanying graph depicts results from a recent year in which there were 8878 male graduates and 8203 female graduates from medical schools in the United States. Does the graph depict the data...
-
Hardin Services Co. experienced the following events in 2016: 1. Provided services on account. 2. Collected cash for accounts receivable. 3. Attempted to collect an account and, when unsuccessful,...
-
(a) The following complexes each possess one of the structures listed in Table 19.4. Use the point group to deduce each structure: [ZnCl 4 ] 2 (T d ); [AgCl 3 ] 2 (D 3h ); [ZrF 7 ] 3 (C 2v ); [ReH 9...
-
Interactions between DNA and metal complexes are the basis for the use of square planar platinum(II)-containing anti-cancer drugs. (a) Explain how the interaction of right-handed DNA with chiral...
-
(a) In the solid state, Fe(CO) 5 possesses a trigonal bipyramidal structure. How many carbon environments are there? (b) Explain why only one signal is observed in the 13 C NMR spectrum of solutions...
-
What are the key challenges in designing fault-tolerant operating systems capable of providing seamless continuity of service in the face of hardware failures, software errors, and malicious attacks,...
-
Danielle is a contractor and operates her own home remodeling business. She just completed two projects, revealed the finished products to her clients, and submitted the invoices to them. At this...
-
please as a reference take any Indian movie or tv show if possible Topics: This course is examining concepts of male and female in the west, and so your research topic should be something about...

Study smarter with the SolutionInn App


