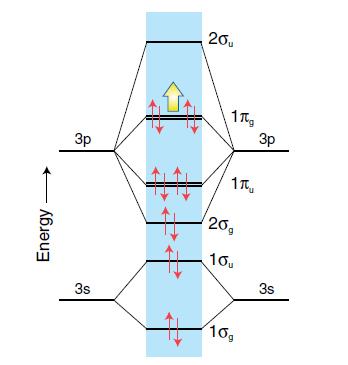
Use the molecular orbital diagram in Fig. 17.5 to determine the bond order of the Br 2
Question:
Use the molecular orbital diagram in Fig. 17.5 to determine the bond order of the Br2+ ion. Will the Br–Br bond be longer or shorter than that in the Br2 molecule?
Figure 17.5.
Transcribed Image Text:
Energy Зр 3s 20₁ 1 1 20g 10, 10g 3p 3s
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (5 reviews)
Bond order is calculated by the formula 12number of electrons in bonding ...View the full answer

Answered By

User l_1028164
I'm masters in chemistry and I've pursued my post graduation from central university of Haryana. I'm fascinated about teaching and I've three years of experience as subject matter expert.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Inorganic Chemistry
ISBN: 9780198768128
7th Edition
Authors: Mark Weller, Tina Overton, Jonathan Rourke
Question Posted:
Students also viewed these Sciences questions
-
The oxygenoxygen bond in O2+ is 112 pm and in O2 is 121 pm. Explain why the bond length in O2+ is shorter than in O2. Would you expect the bond length in O2 to be longer or shorter than that in O2?...
-
Consider the Molecular Orbital diagram for the ion H2 (An H2 ion with 3 e"). Predict the bond order. 2.0 0.5 1.0 1.5 Submit Answer Tries 0/2 Consider the following statements. v) Will the ion be...
-
The duration of copyright increased under U.S. law in several steps since the eighteenth century until it reached the life of the author plus 70 years. Suppose that a writer completes a novel at age...
-
Regulation imperial soccer fields have a length of 91 meters and a width of 39 meters. Show that during a friendly match there are always two players that are not farther from each other than 19...
-
Identify the accounts listed in QS 4-9 that would be included in a post-closing trial balance.
-
The operations manager at a light emitting diode (LED) light bulb factory needs to estimate the mean life of a large shipment of LEDs. The manufacturers specifications are that the standard deviation...
-
Choose a country from three of the regions presented in Table 6.7. Using the Internet, collect as much information as you believe is needed to identify the potential for market segments based on age,...
-
Fibertek, Inc., computed a pretax financial income of $40,000 for the first year of its operations ended December 31, 2011. Included in financial income was $25,000 of nondeductible expenses, $22,000...
-
3. Write a test program which has a static method that takes a shape argument and displays information about it (its radius, its area and volume, if applicable). Writing this to display information...
-
The nature of the sulfur cycle in ancient times has been investigated (J. Farquhar, H. Bao, and M. Thiemen, Science, 2000, 289, 756). What three factors influence the modern-day cycle? When did the...
-
Rank the following species from the strongest reducing agent to the strongest oxidizing agent: SO 4 2 , SO 3 2 ,O 3 SO 2 SO 3 2 .
-
1. What does Stevenson think about the usefulness of science in resolving ethical disputes? 2. How are ethical claims used to create an influence? 3. What does Stevenson mean when he says that...
-
In small community of Swan River, Manitoba, what non-financial co-operatives organizations are you aware of? What functions do these co-operatives perform? What other opportunities and benefits do...
-
What are public goods and services? How do governments produce public goods and services? What is the impact of taxation on the type of things that government can provide to the people? How does the...
-
an achieved status that hold. How did this achieved status become a part of identity? please add refrences
-
Consider the mapping zz. Plot the image of the region {z = C | Re(z), Im(z) [0, 1]}.
-
The most terrifying words For example, what about fairness? A free market economy rewards participants according to their ability to work hard, take risks, innovate, create, and ultimately produce...
-
In the Chapter Problem, it was noted that researchers conducted a study to investigate the effects of color on cognitive tasks. Words were displayed on a computer screen with background colors of red...
-
At 31 December 20X9, the end of the annual reporting period, the accounts of Huron Company showed the following: a. Sales revenue for 20X9, $ 2,950,000, of which one- quarter was on credit. b....
-
(a) Write an equation to represent the discharge of an alkaline electrolyte cell containing a Zn anode and BaFeO 4 cathode. (b) The first charge transfer band for [MnO 4 ] occurs at 18320cm 1 , and...
-
When H 2 S is passed into a solution of copper(II) sulfate acidified with H 2 SO 4 , copper(II) sulfide precipitates. When concentrated H 2 SO 4 is heated with metallic Cu, the principal...
-
Dimethyl sulfoxide (DMSO) reacts with cobalt(II) perchlorate in EtOH to give a pink compound A which is a 1 : 2 electrolyte and has a magnetic moment of 4.9 B . Cobalt(II) chloride also reacts with...
-
ruta Inc. provided the following data: June July August September Net Sales 1,274,000 676,000 1,020,000 780,000 Purchases of merchandise (includes freight-in) 760,000 65,0000 700,000 560,000...
-
1. using python input() function to ready both integer and float values such as, x = 7 and h = 0.05 2. Write a python function for f(x + h) 3. Write a python function for f(x) 4. write a python...
-
The app has three sections, a list of tags to filter events, a list of upcoming events, and the user's agenda. The tag list displays the set of tags returned from the backend that are extracted from...

Study smarter with the SolutionInn App


