Using hardsoft concepts, which of the following reactions are predicted to have an equilibrium constant greater than
Question:
Using hard–soft concepts, which of the following reactions are predicted to have an equilibrium constant greater than 1? Unless otherwise stated, assume gas-phase or hydrocarbon solution and 25°C.
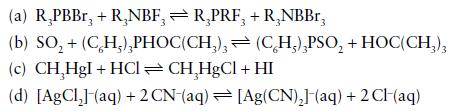
Transcribed Image Text:
(a) R₂PBBr, + R,NBF, R₂PRF, + R₂NBBr, 3 (b) SO₂ + (CH₂),PHOC(CH₂), (c) CH₂HgI + HCl =CH₂HgCl + HI (d) [AgCl₂] (aq) + 2 CN-(aq) [Ag(CN)₂] (aq) + 2CH(aq) (CH₂),PSO₂ + HOC(CH3)3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (3 reviews)
The prediction of equilibrium constants based on hardsoft acidbase concepts involves considering the ...View the full answer

Answered By

Collins Njuguna
I graduated from Maseno University with a Bachelor of Science in Applied Statistics. After graduation, I started tutoring students in mathematics. My experience in mathematics education is extensive and varied. I have taught a wide range of topics, including algebra, geometry, trigonometry, calculus, statistics, probability, and computer science. I have also worked with students of all ages and backgrounds, from elementary school to college.
My teaching method is based on the idea of hands-on learning. I believe that students learn best when they are actively engaged in the learning process, so I focus on giving students the tools they need to explore the material on their own. I also emphasize the importance of practice and review, as these are essential for mastering math concepts.
I have also developed several online and in-person courses on mathematics. My courses are designed to help students learn mathematics in an efficient and comprehensive way, and I use a variety of activities and exercises to ensure that my students are engaged and motivated.
Overall, my passion for mathematics and teaching has allowed me to be a successful tutor and educator in the field. I am confident that my experience will help your students master the mathematics they need to succeed.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Inorganic Chemistry
ISBN: 9780198768128
7th Edition
Authors: Mark Weller, Tina Overton, Jonathan Rourke
Question Posted:
Students also viewed these Sciences questions
-
In the diagram, ABC ~ DEF. Find the scale factor of ABC to DEF. D E 8 2|5 10//2 9 F A 22 B 19 C
-
KYC's stock price can go up by 15 percent every year, or down by 10 percent. Both outcomes are equally likely. The risk free rate is 5 percent, and the current stock price of KYC is 100. (a) Price a...
-
1. How strong are the competitive forces confronting J. Crew in the market for specialty retail? Do a [Michael Porter] five-forces analysis to support your answer. (see chapter 3 in the textfor...
-
1. The standard price per unit of materials is used in the calculation of which of the following variances? Materials price variance Materials quantity variance a NO NO b NO YES c YES NO d YES YES 2...
-
The Westwood Management Association held its annual public relations luncheon in April 2013. 1Based on the previous years results, the organization allocated $25,200 of its operating budget to cover...
-
Rochelle is a partner in Megawatt Partnership. For 2016, her schedule K-1 from the partnership reported the following share of partnership items: Ordinary...
-
Should the requirements of the UCC be subject to the application of reliance theories? Go back and review the facts in Case 21-3 about the coal contract. Should silence followed by contract execution...
-
On October 1, White Way Stores Inc. is considering leasing a building and purchasing the necessary equipment to operate a retail store. Alternatively, the company could use the funds to invest in $...
-
What type(s) of error(s) occurs in the following deduction? Briefly justify your answer. 4 = 2+3 4(4)=4(2+3) 4(4)-5=4(2+3)-20 16-5=20-20 11 = 0
-
The aqueous solution pK a values for HOCN, H 2 NCN, and CH 3 CN are approximately 4, 10.5, and 20 (estimated), respectively. Explain the trend in these cyano derivatives of binary acids and compare...
-
An article by Krossing and co-workers (J. Am. Chem. Soc., 2006, 128, 13427) explains the behaviour of ionic liquids in terms of a thermodynamic cycle approach. Describe the principles that are...
-
What are some of the common marketing options for the consumer market? Think of a product and discuss how you would mix and match the communication options for that product.
-
Zack Manufacturing Company makes a product that is processed through two departments: cutting and final. All materials are added in the first department. During the month of March 2019, the company...
-
Assume market rate of return of 14%, risk-free rate of 4% and beta of 0.98 for BEA Systems. Assume also that BEA will continue to grow at its recent average annual growth rate of 9% for indefinite...
-
Today, numerous painkillers and cold and flu therapies are available over the counter. In general, people tend to buy these OTCs without consulting a doctor or pharmacist. Do you feel people are...
-
What is the ending balance of the current year deferred tax liability?
-
As we look to the future of sport, why do you think that soccer is less popular in the United States? Will it eventually catch on? If you were the person in charge of choosing a sport to use in a...
-
The stable isotopes of iron are 54Fe, 56Fe, 57Fe, and 58Fe. The mass spectrum of iron looks like the following: 100 E 80 60 91.75 3 20 585 212 0.28 54 56 57 58 Mass sumber
-
Explain the differences and similarities between fringe benefits and salary as forms of compensation.
-
When NaCN dissolves in water, the resulting solution is basic. Account for this observation given that pK a for HCN is 9.31.
-
(a) Discuss the factors that contribute towards KCl being a readily soluble salt (35 g per 100 g H 2 O at 298 K). (b) Develop your answer to part (a) by using the following data: hyd H(K + , g) =...
-
Potassium chromate is used as an indicator in titrations for the determination of chloride ion. At the end-point of a titration of an aqueous solution of a metal chloride salt (e.g. NaCl) against...
-
Your true height is 70.4 inches. A tape measure that can be read to the nearest tenth of an inch gives your height as 70.5 inches. A second tape measure which can be read to the nearest eighth of an...
-
Question 1 (1 point) Listen A long-term care resident is to take 15 mL of a medication 3 times a day. How many teaspoons (tsp) will this patient need for a 3 day supply of this medication based on...
-
Use the following instructions to conduct the z-test for thepopulation proportion: Instructions Excel Instructions: Hypothesis Testing for thePopulation Proportion p To conduct the z-test for the...

Study smarter with the SolutionInn App


