What isomers would you expect to exist for the platinum(II) compounds: (a) [Pt (HNCHCHMENH)2] Cl, and (b)
Question:
What isomers would you expect to exist for the platinum(II) compounds:
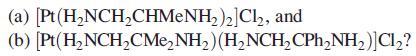
Transcribed Image Text:
(a) [Pt (H₂NCH₂CHMENH₂)2] Cl₂, and (b) [Pt (H₂NCH₂CMе₂NH₂) (H₂NCH₂ CPh₂NH₂)] Cl₂?
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (3 reviews)
To determine the isomers of the platinumII compounds we need to consider the possible arrangements o...View the full answer

Answered By

Abdul Wahab Qaiser
Before working at Mariakani, I volunteered at a local community center, where I tutored students from diverse backgrounds. I helped them improve their academic performance and develop self-esteem and confidence. I used creative teaching methods, such as role-playing and group discussions, to make the learning experience more engaging and enjoyable.
In addition, I have conducted workshops and training sessions for educators and mental health professionals on various topics related to counseling and psychology. I have presented research papers at conferences and published articles in academic journals.
Overall, I am passionate about sharing my knowledge and helping others achieve their goals. I believe that tutoring is an excellent way to make a positive impact on people's lives, and I am committed to providing high-quality, personalized instruction to my students.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
What compounds would you expect to be formed when each of the following ethers is refluxed with excess concentrated hydrobromic acid? (a) (b) (c) (d) (THF) (1,4-dioxane)
-
Fluorination of (R)-2-fluorobutane yields a mixture of isomers with the formula C 4 H 8 F 2 . (a) How many different isomers would you expect to be produced? Write their structures. (b) If the...
-
Which of these isomers would you expect to have the higher boiling point: Explain? CH 3 CH 2 CH 2 OH or CH 3 CH 2 OCH 3
-
Are the marks one receives in a course related to the amount of time spent studying the subject? To analyze this mysterious possibility, a student took a random sample of 10 students who had enrolled...
-
What are the major priorities associated with operations and supply chain strategy? How has their relationship to each other changed over the years?
-
If i s (t) = 7.5e 2t u(t) A in the circuit shown in Fig. 16.40 , find the value of i o (t). i,(t) is(t) 0.5 F 2 relle
-
Consider the following cash flow diagram. What is the value of \(X\) if the internal rate of return is 15 percent? a. \(\$ 246\) b. \(\$ 255\) c. \(\$ 281\) d. \(\$ 290\) 0 $400 X X 1 2
-
Arsenal Electronics is going to construct a new $1.2 billion semiconductor plant and has selected four towns in the Midwest as potential sites. The important location factors and ratings for each...
-
To determine the kinetics (rates) of ozone depletion reactions, chemists perform controlled laboratory studies. In this simulated lab, we will interpret data obtained from such laboratory experiments...
-
Give a brief overview of properties that characterize a d-block metal.
-
By referring to relevant sections earlier in the book, write a brief account of the formation of hydrides, borides, carbides and nitrides of the d-block metals.
-
What is the general name given to biological catalysts?
-
Discuss the role of social influence and group processes in health-related behavior.
-
The default cluster role that provides read-only access is known as the role. 1) read 2) view 3) read-only 4) guest 5) protected
-
What systematic approach or algorithmic technique is employed to execute an inorder traversal within a specified binary tree?"
-
Oppenheimer Bank is offering a 30-year mortgage with an EAR of 6.375%. If you plan to borrow $180,000, what will your monthly payment be? Your monthly payment will be $______(round to nearest cent)
-
James Appliances sells a kitchen appliance set for $12,000. The inventory sold is valued at $7200. The customer pays $1000 down and puts the rest on account. The customer takes possession of...
-
On December 1, 2012, Lavender Manufacturing Company (a corporation) purchased another company's assets, including a patent. The patent was used in Lavender's manufacturing operations; $49,500 was...
-
MgO prevents premature evaporation of Al in a furnace by maintaining the aluminum as Al2O3. Another type of matrix modifier prevents loss of signal from the atom X that readily forms the molecular...
-
Identify which of the following two reactions you would expect to occur more rapidly: (1) addition of HBr to 2-methyl-2- pentene or (2) addition of HBr to 4-methyl-1-pentene. Explain your choice.
-
With current spectroscopic techniques (discussed in Chapters 1517), chemists are generally able to determine the structure of an unknown organic compound in just one day. These techniques have only...
-
Draw the structure of each of the following compounds: a. (R)-3, 3-Dibromocyclohexanol b. (S)-2, 3-Dimethyl-3-pentanol c. (1S, 2S, 4R)-Bicyclo [2.2.1] heptan-2-ol
-
What Behavioral Competency do you see being used by the HR professional in the scenario? What action shows this? Nepotism An HR payroll specialist is in charge of entering payroll data each week and...
-
Calculating total landed cost include cost, freight, duty, and exchange rates. What are other components that are not as obvious that should be considered when determining total landed cost? Is it...
-
What is bank measure? How does the quantity of excavation vary with swell and compaction (be descriptive)? Give an example of how you have been affected by swell/compaction of soil in a project....

Study smarter with the SolutionInn App


