Which of the following ions are diamagnetic: Rationalize your answer. (a) [Co(OH)6]+, (b) [CoF, (c) [NiF, (d)
Question:
Which of the following ions are diamagnetic:
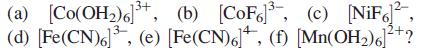
Rationalize your answer.
Transcribed Image Text:
(a) [Co(OH₂)6]³+, (b) [CoF³, (c) [NiF², (d) [Fe(CN)6]³, (e) [Fe(CN)6], (f) [Mn(OH₂)6]²+?
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 25% (4 reviews)
To determine if an ion is diamagnetic or paramagnetic we need to consider its electron configuration ...View the full answer

Answered By

Aketch Cindy Sunday
I am a certified tutor with over two years of experience tutoring . I have a passion for helping students learn and grow, and I firmly believe that every student has the potential to be successful. I have a wide range of experience working with students of all ages and abilities, and I am confident that I can help students succeed in school.
I have experience working with students who have a wide range of abilities. I have also worked with gifted and talented students, and I am familiar with a variety of enrichment and acceleration strategies.
I am a patient and supportive tutor who is dedicated to helping my students reach their full potential. Thank you for your time and consideration.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
can all carbohydrates be digested by humans? explain your answer
-
Write down all the steps of 1) Spanning tree (Prim's & Kruscal) and 2) Topological search (BFS & DFS) algorithms
-
Which of the following ions possess a noble gas electron configuration? (a) K+ (d) O- (b) He+ (e) F- (c) H- (f) Ca2+
-
Why are services important? Why is manufacturing important? What are nonmanufactured goods?
-
Perot Corporation is developing a new CPU chip based on a new type of technology. Its new chip, the Patay2 chip, will take two years to develop. However, because other chip manufacturers will be able...
-
Find the Laplace transform of the signal in Fig. 15.26. f(t) A 25 2 4
-
In 1951, DuPont began using the chemical perfluorooctanoic acid to manufacture Teflon. Due to the dangerous nature of the chemical, DuPont was given special instructions by its supplier to dispose of...
-
A company has three warehouses that supply four stores with a given product. Each warehouse has 30 units of the product. Stores 1, 2, 3, and 4 require 20, 25, 30, and 35 units of the product,...
-
How would you import a relational model design and generate a logical model from a relational model?
-
Deduce possible J values for a 3 F term. What is the degeneracy of each of these J levels, and what happens when a magnetic field is applied? Sketch an energy level diagram to illustrate your answer,...
-
What are the limitations of the RussellSaunders coupling scheme?
-
Vox Company started the period with cash of $22,000, common stock of $12,000, and retained earnings of $10,000. Vox engaged in the following transactions in 2016: Transactions During 2016 1. On...
-
A small ball of mass m and radius r is released from a height h = 3R. The ball rolls without slipping down the incined plane and over the circular track of radius R. Find the speed of the ball at...
-
A block of mass m = 8,9 kg slides on a rough surface and moves toward a spring with a spring constant k = 1004 N/m, as shown in the figure below. When the block is d = 13,1 m away from the spring, it...
-
Photon Corporation has a client who wants to purchase $50,000 of goods on credit. Photon thinks that the client has a 97% probability of paying the $50,000 in 3 months and a 3% chance of a complete...
-
A block of mass m = 13,9 kg is attached to the free end of a light string wrapped around a reel of radius R = 0,38 m and mass M = 7,2 kg. The reel is a uniform disk and fixed on an axis about which...
-
Define an AVL-2 tree to be a BST where each node v satisfies balance(v) {-2,-1,0,1,2}. Let N(h) be the minimum number of nodes in an AVL-2 tree of height h. Prove that h = O(lgn) for the case where...
-
In 2014, Kathleen Tweardy incurs $30,000 of interest expense related to her investments. Her investment income includes $7,500 of interest, $6,000 of qualified dividends, and a $12,000 net capital...
-
Use of the contraceptive Depo Provera appears to triple women's risk of infection with chlamydia and gonorrhea , a study reports today. An estimated 20 million to 30 million women worldwide use Depo...
-
Identify reagents that can be used to achieve each of the following transformations: a. b. c. Br
-
Borane (BH 3 ) is very unstable and quite reactive. Draw a Lewis structure of borane and explain the source of the instability.
-
Propose a plausible synthesis for each of the following transformations: a. b. c. Br Br
-
The objective of this homework is to build a coin counter for a vending machine. The vending machine counts coins in any combination up to 25 cents. 1) Draw a state diagram, you should have states...
-
7. [3 Points] Find the value of the following references based on given information. You may assume that the size of integer type is four bytes and memory address of A[0][0] is 6422212. Given: int...
-
20ns The following table summarizes some relevant parameters relating to the memory system: Cache (SRAM) Access Time DRAM Access Time - best case (see comment below) DRAM Cycle Time - worst case (see...

Study smarter with the SolutionInn App


