With reference to Box 23.2, develop a qualitative bonding scheme for ( 5 -Cp) 2 Fe. Box
Question:
With reference to Box 23.2, develop a qualitative bonding scheme for (η5-Cp)2Fe.
Box 23.2.

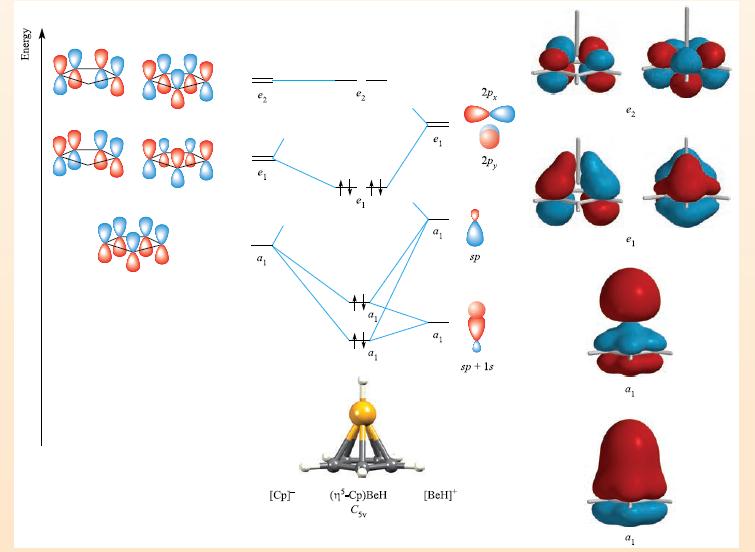
Transcribed Image Text:
THEORY Box 23.2 Bonding in cyclopentadienyl complexes: ³-mode If all five C atoms of the cyclopentadienyl ring interact with the metal atom, the bonding is most readily described in terms of an MO scheme. Once the o-bonding framework of the [Cp] ligand has been formed, there is one 2p, atomic orbital per C atom remaining, and five combinations are possible. The MO diagram below shows the formation of (n-Cp)BeH (Csv), a model compound that allows us to see how the [n³-Cp] ligand interacts with an s-or p-block metal fragment. For the formation of the [BeH]* fragment, we can use an sp hybridization scheme. One sp hybrid points at the H atom and the other points at the Cp ring. Using the methods from Chapter 5, the orbitals of the [BeH] unit are classified as having a, or e, symmetry within the Csv point group. To work out the 7-orbitals of the [Cp] ligand, we first determine how many C 2p, orbitals are unchanged by each symmetry operation in the Cs, point group (Appendix 3). The resultant row of characters is: 2C5 2C3 E 5 00 Say 1 This row can be obtained by adding the rows of characters for the A₁, E₁ and E₂ representations in the C5, character table. Thus, the five 7-orbitals of [Cp] possess a₁, e, and e₂ symme- tries. By applying the methods described in Chapter 5, the wavefunctions for these orbitals can be determined. The orbi- tals are shown schematically on the left-hand side of the diagram. The MO diagram is constructed by matching the symmetries of the fragment orbitals. Mixing can occur between the two a₁ orbitals of the [BeH] fragment. Four bonding MOs (a, and e₁) result. The e₂ [Cp] orbitals are non-bonding with respect to Cp-BeH interactions. (Anti- bonding MOs have been omitted from the diagram.) Eight electrons are available to occupy the a₁ and e₁ MOs. Repre- sentations of the a₁, e, and e₂ MOs are shown at the right- hand side of the figure: the e, set possesses Be-C bonding character, while both a MOs have Be-C and Be-H bonding character. Bonding in cyclopentadienyl complexes of d-block metals (see Chapter 24) can be described in a similar manner but must allow for the participation of metal d-orbitals.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (4 reviews)
Here is a qualitative bonding scheme for 5Cp2Fe 1 Cyclopentadienyl Ligands Cp Each cyclopentadienyl ...View the full answer

Answered By

Umber Talat
I am providing full time mentoring and tutoring services in Business Finance, Contemporary issue in Global Economy, Quantitative Techniques, Principles of Marketing, strategic marketing, International Marketing, Organizational Behavior (OB), Consumer Behavior, Sales Force Management, Strategic Brand Management, Services Marketing, Integrated Marketing Communication (IMC), Principles of Management, General Management, Strategic Management, Small and Medium Enterprise Management, Innovation Management, Change Management, Knowledge Management, Strategic Planning, Operations Management, Supply Chain Management, Logistics Management, Inventory management, Total Quality Management (TQM), Productions Management, Project Management, Production Planning, Human Resource Management (HRM), Human Resource Development, Strategic HRM, Organizational Planning, Performance and Compensation Management, Recruitment and Selection, Organizational Development, Global Issues in Human Resource Management, Retail Marketing, Entrepreneurship, Entrepreneurial Marketing, International Business, Research Methods in Business, Business Communication, Business Ethics.
4.70+
158+ Reviews
236+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Write a literature review for your study. See below for an example of a literature review. Your literature review should provide both analysis and synthesis of previous studies as related to the...
-
List three specific parts of the Case Guide, Objectives and Strategy Section (See below) that you had the most difficulty understanding. Describe your current understanding of these parts. Provide...
-
A manufacturer of network computer server systems is interested in improving its customer support services. As a first step, its marketing department has been charged with the responsibility of...
-
Erin McQueen purchased 50 shares of BMW, a German stock traded on the Frankfurt Exchange, for 64.5 euros () per share exactly 1 year ago when the exchange rate was 0.67 /US$. Today the stock is...
-
Jack Werth, the owner-president of Computer Services Company, is unfamiliar with the statement of cash flows that you, as his accountant, prepared. He asks for further explanation. Instructions Write...
-
Use the Factor Theorem to prove that x c is a factor of x n c n for any positive integer n.
-
Brian Rafferty ingested finasteride, a drug prescribed to him to treat an enlarged prostate. Finasteride is a generic version of Proscar, a drug manufactured by Merck. Some time after he started to...
-
Jefferson County General Fund began the year 2012 with the following account balances: During 2012, Jefferson experienced the following transactions: 1. The budget was passed by the County...
-
Research articles on Online Analytic Processing (OLAP) and Online Transaction Processing (OLTP). Next, compare and contrast the key similarities and differences between Online Analytical Processing...
-
(a) Os 5 (CO) 18 has a metal framework consisting of three edge-sharing triangles (a raft structure). Show that the valence electron count for this raft is consistent with the number available. (b)...
-
In Section 24.8, we stated that the distribution of the products in Fig. 24.18 is consistent with the migration of the Me group, and not with a mechanism that involves movement of the inserted CO....
-
Figure shows a cross section of a hollow cylindrical conductor of radii a and b, carrying a uniformly distributed current t. (a) Show that the magnetic field magnitude B(r) for the radial distance r...
-
X43X 10 = 0; find X.
-
Solve for x. Inx - In 3+ In (x-8) X = 0 X No solution 3
-
Let f(x) = -10 tan(x) 12 sin(x) 7 cos(x). What is the slope of the tangent line to the curve y = f(x) at x = 4 ?
-
Consider a member method that prints the keys of a BST in a decreasing order, complete the following code by choosing the correct answer: public void printReverse() { 1. ... } private void...
-
Let lim(x) = 3 and limg(x) = 1. x 5 lim [f(x)+g(x)]. x5
-
A number of years ago, Kay acquired an interest in a partnership in which she is not a material participant. Kays basis in her partnership interest at the beginning of 2013 is $40,000. Kays share of...
-
Sundial Technologies produces and sells customized network systems in New Brunswick. The company offers a 60-day, all software and labor-and an extra 90-day, parts-only- warranty on all of its...
-
Identify the major and minor products for the E2 reaction that occurs when each of the following substrates is treated with a strong base: a. b. c. d. e. f. g. h. Br
-
Identify an alkyl halide that could be used to make the following alkene:
-
When menthyl chloride is treated with a strong base, only one elimination product is observed. Yet, when neomenthyl chloride is treated with a strong base, two elimination products are observed. Draw...
-
Azie sells 1,500 carpets a year during the last MCO at an average price per carpet of RM499. The ordering cost per unit is RM13. If the economic order quantity is 100 units, calculate the total...
-
Cik Dah's sells 1,300 fancy cupcakes a year during the last MCO at an average price per cupcake of RM500. The carrying cost per unit is RM15.50. The company orders 60 fancy cupcakes at a time and has...
-
Your next-door neighbor, Peter Gittins, having finished sending his children to college, has decided he needs to get serious about saving for retirement. He hopes to retire in six years and, though...

Study smarter with the SolutionInn App


