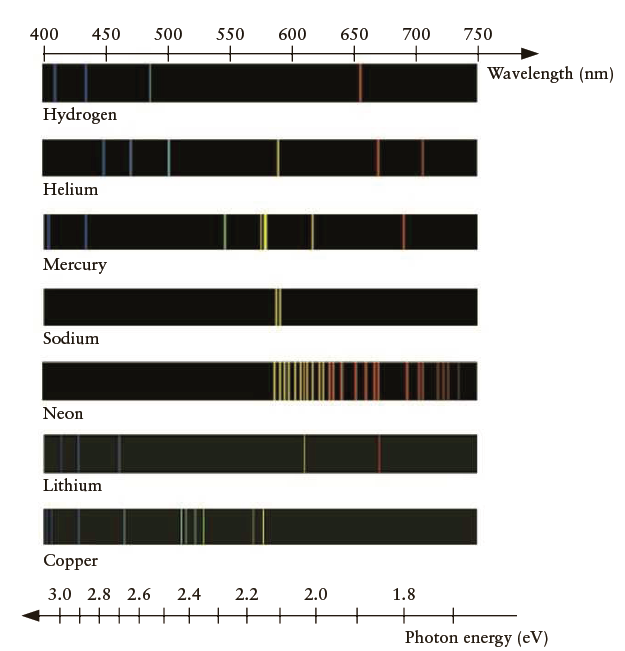
Compare the emission spectra of the elements hydrogen and helium (Figure 10.12). Which element emits photons of
Question:
Compare the emission spectra of the elements hydrogen and helium (Figure 10.12). Which element emits photons of red light that have the higher energy?
Figure 10.12
Transcribed Image Text:
650 750 400 450 500 550 600 700 Wavelength (nm) Hydrogen Helium Mercury Sodium Neon Lithium Copper 2.0 1.8 3.0 2.8 2.6 2.4 2.2 + + Photon energy (eV)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (10 reviews)
The red line in the helium spectrum is closer to ...View the full answer

Answered By

Ajeet Singh
Hi there! Are you looking for a committed, reliable, and enthusiastic tutor? Well, teaching and learning are more of a second nature to me, having been raised by parents who are both teachers. I have done plenty of studying and lots of learning on many exciting and challenging topics. All these experiences have influenced my decision to take on the teaching role in various capacities. As a tutor, I am looking forward to getting to understand your needs and helping you achieve your academic goals. I'm highly flexible and contactable. I am available to work on short notice since I only prefer to work with very small and select groups of students. Areas of interest: Business, accounting, Project management, sociology, technology, computers, English, linguistics, media, philosophy, political science, statistics, data science, Excel, psychology, art, history, health education, gender studies, cultural studies, ethics, religion. I am also decent with math(s) & Programming. If you have a project you think I can take on, please feel welcome to invite me, and I'm going to check it out!
5.00+
4+ Reviews
24+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Physics questions
-
Identifying Isotopes by Spectra Different isotopes of the same element emit light at slightly different wavelengths. A wavelength in the emission spectrum of a hydrogen atom is 656.45 nm; for...
-
Laser atomic fluorescence excitation and emission spectra of sodium in an air-acetylene flame are shown here. In the excitation spectrum, the laser (bandwidth 0.03 nm) was scanned through various...
-
Hydrogen sulfide is composed of two elements: hydrogen and sulfur. In an experiment, 6.500 g of hydrogen sulfide is fully decomposed into its elements. (a) If 0.384 g of hydrogen is obtained in this...
-
What are the explanations given for slow progress towards full acceptance of ISAs?
-
Discuss the reasons behind the new expansionism that characterized US foreign policy in 1890's.
-
This case, based on Rainmaker Entertainment Inc.'s balance sheet on My Accounting Lab, will familiarize you with some of the assets and liabilities of this actual company. Answer these questions,...
-
What departments report to (a) the vice-president of manufacturing and (b) the controller in the expenditure cycle?
-
The following questions refer to the CRT Technologies project selection example presented in this chapter. Formulate a constraint to implement the conditions described in each of the following...
-
Consider a cake pan that is 12in x 12in x 3.0in. (a) How much water(in mL) can the cake pan hold? (b) How high(in mm) would the cake pan be filled if you added 0.50 kg of water to it? The density of...
-
A company needs to hold a stock of item X for sale to customers. Although the item is of relatively small value per unit, the customers' quality control requirements and the need to obtain...
-
Discuss what is meant by the term ionization. Give two ways by which an atom might acquire enough energy to become ionized.
-
If an astronomer examines the emission spectrum from luminous hydrogen gas that is moving away from Earth at a high speed and compares it to a spectrum of hydrogen seen in a laboratory on Earth, what...
-
The following information is available for Bergstrom Inc. and Sineann Inc. at December 31, 2016: Required a. What is the accounts receivable turnover for each of the companies for 2016? b. What are...
-
The following data have been extracted from the records of Puzzle Incorporated: Production level, in units Variable costs Fixed costs Mixed costs Total costs Required: a. Calculate the missing costs....
-
Over the past 3 years, Citrus Health (or "Hospital") has experienced a higher than usual amount of medical malpractice and wrongful death claims. One neurosurgeon experienced five paralysis cases...
-
Add more relevant law and law sections. Also add relevant case law. Issue: Is Earth Care, in its capacity as a GreenCo shareholder, able to contest the contact's validity on the grounds of violation...
-
Discuss the role of systems biology in understanding and optimizing complex bioprocesses. How do computational models and omics data contribute to the prediction and enhancement of bioprocess...
-
Bonita, Inc. decided to implement the activity-based costing approach and was quite successful in its use. However, the controller is wondering if instead of only two activity cost pools, they should...
-
The correlation chapter was about understanding ____ between variables; this chapter, on regression, is about ____ one ____ from another ____.
-
A bubble-point liquid feed is to be distilled as shown in Figure. Use the Edmister group method to estimate the mole-fraction compositions of the distillate and bottoms. Assume initial overhead and...
-
Digesting fat produces 9.3 food calories per gram of fat, and typically 80% of this energy goes to heat when metabolized. (One food calorie is 1000 calories and therefore equals 4186 J.) The body...
-
An average sleeping person metabolizes at a rate of about 80 W by digesting food or burning fat. Typically, 20% of this energy goes into bodily functions, such as cell repair, pumping blood, and...
-
A certain heat engine operating on a Carnot cycle absorbs 150 J of heat per cycle at its hot reservoir at 135C and has a thermal efficiency of 22.0%. (a) How much work does this engine do per cycle?...
-
You decide to travel by car for your holiday visits this year. You leave early in the morning to avoid congestion on the roads. This enables you to drive at a comfortable speed of v = 67.1 mph for t...
-
The figure below shows three Gaussian surfaces and the electric flux through each. If q = 38.68nC. = A -gleo (91) What is the charge of q? Submit Answer Tries 0/10 What is the charge of 92? Submit...
-
Suppose the normalized pulse shape, h(t), shown below, is used to generate the pulse train v(t) = vnhr(t - nT) N=- where {n} are the transmitted symbols, with each Un selected independently from {-1,...

Study smarter with the SolutionInn App


