For the ammonia synthesis reaction written: With 0.5 mol N 2 and 1.5 mol H 2 as
Question:
For the ammonia synthesis reaction written:
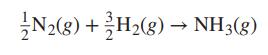
With 0.5 mol N2 and 1.5 mol H2 as the initial amounts of reactants and with the assumption that the equilibrium mixture is in the ideal gas state, show that:
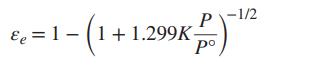
Transcribed Image Text:
N2(8) + H₂(g) → NH3(g)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 33% (3 reviews)
It seems like you want to analyze the ammonia synthesis reaction and its equilibrium using the given ...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Introduction To Chemical Engineering Thermodynamics
ISBN: 9781260597684
9th International Edition
Authors: J.M. Smith, Mark Swihart Hendrick C. Van Ness, Michael Abbott
Question Posted:
Students also viewed these Engineering questions
-
For the ammonia synthesis reaction written: with 0.5 mol N 2 and 1.5 mol H 2 as the initial amounts of reactants and with the assumption that the equilibrium mixture is an ideal gas, show that: N2{g)...
-
During an experiment with the Haber process, a researcher put 1 mol N2 and 1 mol H2 into a reaction vessel to observe the equilibrium formation of ammonia, NH3. When these reactants come to...
-
Air flows with negligible friction through a 4-indiameter duct at a rate of = lbm/s. The temperature and pressure at the inlet are T1 = 800 R and P1 = 30 psia, and the Mach number at the exit is Ma2...
-
The concept of beta is most closely associated with: a. Correlation coefficients. b. Mean-variance analysis. c. Nonsystematic risk. d. Systematic risk.
-
For the situation and data described in Exercise 17.17, perform conventional multiple regression and stepwise regression analyses on the data, then compare the results of the two approaches. Use...
-
Analyze the fuel consumption data in Table B. 18 for multicollinearity. y X2 X3 X4 xs X6 X7 Xg 343 0 52.8 811.7 2.11 220 261 87 1.8 356 1 52.8 811.7 2.11 220 261 87 1.8 344 0 50.0 821.3 2.11 223 260...
-
Wagner Manufacturing estimated its product costs and volume of production for 2012 by quarter as follows. Wagner Company sells a souvenir item at various resorts across the country. Its management...
-
Direct materials Average Cost per Unit $ 7.80 Direct labor $ 4.80 Variable manufacturing overhead $ 2.30 .Fixed manufacturing overhead $ 5.80 Fixed selling expense $ 4.30 Fixed administrative expense...
-
Consider the charity auction. In many charity auctions, altruistic celebrities auction objects with special value for their fans to raise funds for charity. Madonna, for example, held an auction to...
-
Carbon black is produced by the decomposition of methane: For equilibrium at 650C and 1 bar, (a). What is the gas-phase composition if pure methane enters the reactor, and what fraction of the...
-
A system formed initially of 2 mol CO 2 , 5 mol H 2 , and 1 mol CO undergoes the reactions: Develop expressions for the mole fractions of the reacting species as functions of the reaction coordinates...
-
In each chapter of this book, we use a database for a fictitious company, Performance Lawn Equipment (PLE), within a case exercise for applying the tools and techniques introduced in the chapter. 33...
-
State the appointment procedure and terms of office of CAG of India.
-
Information and means of information are by no means equivalent terms. Explain.
-
State the main features of departmental undertakings.
-
How and in what way does verification of assets and liabilities differ from vouching?
-
What are the Acts relevant for bank audit?
-
Polar Containers makes high-end coolers for camping. The total task time needed to make a cooler is 360 seconds, with the longest individual task taking 50 seconds. Polar Containers would like to set...
-
Respond to the ethical judgments required based on the following scenarios. Scenario 1. Assume you have collected a sample using MUS and that you have evaluated that sample to calculate a total...
-
Use the UNIFAC model to predict P-x-y data at 90C and x 1 , = {0, 0.1, 0.3, 0.5, 0.7, 0.9, 1.0} for propanoic acid + water. Fit the UNIQUAC model to the predicted P-x-y data and report your UNIQUAC a...
-
(a) Rearrange Eqn. 13.22 to obtain Eqn. 13.23. (b) Use Eqns. 13.16 and 13.18 in Eqn. 13.17 and perform the integration to obtain Eqn. 13.19. (c) Use Eqns. 13.16 and 13.31 in Eqn. 13.17 and perform...
-
The energy equation for mixtures can be written for polymers in the form: By analogy to the development of the Scatchard-Hildebrand theory, this can be rearranged to: where N di = degree of...
-
The measures of central tendency discussed in this chapter are designed to reveal information about the center of a distribution of values. Measures of dispersion provide information about the spread...
-
Assume that you won the Powerball jackpot. You were given the choice between collecting $758.7 million over 30 years at the beginning of each year, or $480.5 million right away. The current maximum...
-
Have the M&A activities of the firm had a positive or negative effect on the firm's long-term position in its product market areas?

Study smarter with the SolutionInn App


