One mole of an ideal gas with constant heat capacities undergoes an arbitrary mechanically reversible process. Show
Question:
One mole of an ideal gas with constant heat capacities undergoes an arbitrary mechanically reversible process. Show that:
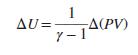
Transcribed Image Text:
AU=- 7-14(PV)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 62% (8 reviews)
To begin we can use the first law of thermodynamics which states that the change in internal energy ...View the full answer

Answered By

Morris Maina
I am a professional teaching in different Colleges and university to solved the Assignments and Project . I am Working more then 3 year Online Teaching in Zoom Meet etc. I will provide you the best answer of your Assignments and Project.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Introduction To Chemical Engineering Thermodynamics
ISBN: 9781259696527
8th Edition
Authors: J.M. Smith, Hendrick Van Ness, Michael Abbott, Mark Swihart
Question Posted:
Students also viewed these Engineering questions
-
An ideal gas with constant heat capacities undergoes a change of state from conditions T1, P1 to conditions T2, P2. Determine (H (J mol-1) and (S (J mol-1 K-1) for one of the following cases. (a) T1...
-
One mole of an ideal gas with heat capacity Cv goes through a process in which its entropy S depends on T as S = a/T, where a is a constant. The gas temperature varies from T1 to T2. Find: (a) The...
-
One mole of an ideal gas with the adiabatic exponent y goes through a polytropic process as a result of which the absolute temperature of the gas increases T-fold. The polytropic constant equals n....
-
Choose a company or product that you have been noticing or admiring. Develop a plan to advertise this product that involves the United States and at least one other country, and three forms of media....
-
Is it possible for a small business owner to take an active role in social responsibility? What are some ways that a small business (for example, a family business or a local business you are...
-
For the figure shown below, find the 4x4 homogeneous transformation matrices A and A for i=1, 2, 3, 4, 5 0 0 n. a, Px -1 e+c n, F = S, a, Py r. -1 a - d | b n. a. . 125 0 0 1 1 e a Yo -Can you find...
-
List and distinguish the three views of structural modeling in the context of an example.
-
Visit the AICPAs website (www.aicpa.org), under Research Standards. On the left side of the page click Code of Professional Conduct. Find Section 100 of the Code of Professional Conduct. Research the...
-
What is Sales Force Automation Function inside the Sales Marketing System. Describe Brielfy.
-
Evaluate HSBC's recent business and marketing shift. How do you think its current ad campaign and tagline, "HSBC helps you unlock the world's potential," resonate with its key consumers?
-
A substance for which is a constant undergoes an isothermal, mechanically reversible process from initial state (P 1 , V 1 ) to final state (P 2 , V 2 ), where V is molar volume. (a) Starting with...
-
Estimate: (a) The mass of ethane contained in a 0.15 m 3 vessel at 60C and 14,000 kPa. (b) The temperature at which 40 kg of ethane stored in a 0.15 m 3 vessel exerts a pressure 20,000 kPa.
-
Write down a formula for the inverse of a Jordan block matrix.
-
Rose Limited operates a small chain of retail shops that sell high-quality teas and coffees. Approximately half of sales are on credit. Abbreviated and unaudited financial statements are given below....
-
Refer to Figure 17.2. Assume the price of virgin materials is p 2 . Identify the level of total materials on the graph, the level of recyclable materials and virgin materials used via curve S 1 ....
-
Threads Limited manufactures nuts and bolts, which are sold to industrial users. The abbreviated financial statements for 2009 and 2010 are as follows: Dividends were paid on ordinary shares of...
-
Explain how the same dollar emission charge satisfies the equimarginal principle while the same percent emissions reduction (equiproportional) results in higher societal costs.
-
Terry bought a house for 65,000 on 1 October 2000 and occupied the house as his principal private residence. He lived in the house until 1 October 2006 when he went to stay with relatives in...
-
(a) Determine whether the polynomials x2 + 1, x2 - 1, x2 + x + I span P(2). (b) Do x3 - I, x2 + 1, x - 1, 1 span P(3)? (c) What about x3, x2 + 1, x2 - x, x + 1?
-
Suppose the S&P 500 futures price is 1000, = 30%, r = 5%, = 5%, T = 1, and n = 3. a. What are the prices of European calls and puts for K = $1000? Why do you find the prices to be equal? b. What...
-
Determine a good estimate for the compressibility factor Z of saturated hydrogen vapor at 25 K and 3-213 bar. For companion, an experimental value is Z = 0.7757.
-
Five kilograms of liquid carbon tetrachloride undergo a mechanically reversible, iso- haric change of state at I bar during which the temperature changes from 0(C to 20(C. Determine (V1, W, , (H1,...
-
Five kilograms of liquid carbon tetrachloride undergo a mechanically reversible, iso- haric change of state at I bar during which the temperature changes from 0(C to 20(C. Determine (V1, W, , (H1,...
-
Include your strategies for capitalizing on your strengths and overcoming your weaknesses in order to become successful in your work related goals
-
when Recognizing and Identifying Child Abuse and Neglect, Include some strategies for capitalizing on strengths and overcoming weaknesses in order to become successful in your work-related goals?
-
What can be the most beneficial when learning about Recognizing and Identifying Child Abuse and Neglect? How can Recognizing and Identifying Child Abuse and Neglect will assist you in achieving...

Study smarter with the SolutionInn App


