The following is the trial balance of A. Evans as at 30 June 20X9: Additional information 1.
Question:
The following is the trial balance of A. Evans as at 30 June 20X9:
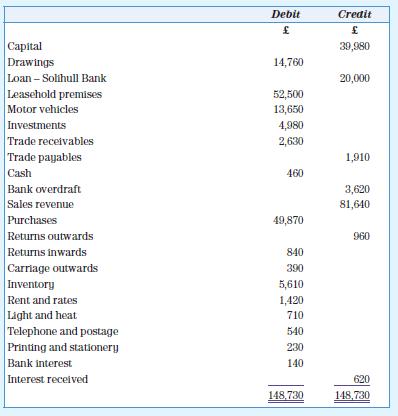
Additional information
1. The inventory at 30 June 20X9 has been valued at £4,920.
2. The bank loan is repayable on 1 June 20Y1.
Required
Prepare a statement of profit or loss for the year ended 30 June 20X9 and a statement of financial position as at that date (publishable format).
Capital Drawings Loan-Solihull Bank Leasehold premises Motor vehicles Investments Trade receivables Trade payables Cash Bank overdraft Sales revenue Purchases Returns outwards Returns inwards Carriage outwards Inventory Rent and rates Light and heat Telephone and postage Printing and stationery Bank interest Interest received Debit £ 14,760 52,500 13,650 4,980 2,630 460 49,870 840 390 5,610 1,420 710 540 230 140 148,730 Credit £ 39,980 20,000 1,910 3,620 81,640 960 620 148,730
Step by Step Answer:
Statement of Profit or Loss for the year ended 30 June 20X9 Income Sales Revenue 81640 Interest Rece...View the full answer



Introduction To Financial Accounting
ISBN: 9781526803009
9th Edition
Authors: Anne Marie Ward, Andrew Thomas
Related Video
A trial balance is a list of all the general ledger accounts contained in the ledger of a business. This list will contain the name of each nominal ledger account and the value of that nominal ledger balance. Each nominal ledger account will hold either a debit balance or a credit balance
Students also viewed these Business questions
-
The trial balance for Eureka Ltd as at 30 June 2012 (before calculation of income tax) is as follows: Note: this is the first year of operation of Eureka Ltd. DR CR $000 $000 Sales Revenue 1235 Cost...
-
If an organization has an average daily gross patient service revenue $230,000 along with 120 patients preregistered encounters, 150 scheduled encounters, and $100,000 in gross dollars in discharged,...
-
The trial balance of Jess Private, a sole trader, for the financial year ended 30 September 2022 is provided below: Jess Private's Trial Balance on the year ended 30 September 2022 Dr (E) Cr ()...
-
You are to show the journal entries necessary to record the following items which occured in 2012: (a) May 1 Bought a van on credit from Deedon Garage for 6,000. (b) May 3 A debt of 100 owing from P....
-
Verify that the butterfly spread in Figure 3.14 can be duplicated by the following transactions (use the option prices in Table 3.4): In Figure 3.14 a. Buy 35 call, sell two 40 calls, buy 45 call. b....
-
For phenanthrene, the measured lifetime of the triplet state ( p ) is 3.3 s, the fluorescence quantum yield is 0.12, and the phosphorescence quantum yield is 0.13 in an alcoholether glass at 77 K....
-
The ideal column has a weight \(w\) (force/length) and is subjected to the axial load \(\mathbf{P}\). Determine the maximum moment in the column at midspan. \(E I\) is constant. Establish the...
-
1. Most aspects of foreign culture, like language, religion, gender roles, and problem-solving strategies, are hard for the casual observer to understand. In what ways do Hollywood movies affect...
-
In the figure an electron (e) is to be released from rest on the central axis of a uniformly charged disk of radius R. The surface charge density on the disk is +4.27 C/m. What is the magnitude of...
-
Bamboo Limited is a highly specialist firm of central heating suppliers operating exclusively in the textiles industry. It has recently been asked to tender for a contract for a prospective customer....
-
Brian Ltd starts selling footballs in 20X8. Although each ball looks the same, the unit cost of manufacture (which is done in batches) has fluctuated during the period. Details of the costs are as...
-
What do you understand by the term irrecoverable debts? In what circumstances might a debt be treated as irrecoverable?
-
In MiniCase 10.2, we found that the mean methane gas emissions for the two pneumatic controller manufacturers were not equal. When choosing formula 10.3 to calculate the t statistic, we assumed that...
-
Which of the following are examples of molecular changes that can have an epigenetic effect on gene expression? a. Chromatin remodeling b. Covalent histone modification c. Localization of histone...
-
The site(s) on a ribosome where tRNA molecules may be located include a. the A site. c. the E site. b. the P site. d. all of the above.
-
Which of the following statements about paramutation is false? a. A paramutagenic allele can alter the expression of paramutable allele. b. A paramutagenic allele has a lower level of expression than...
-
Heterochromatin is characterized by which of the following? a. Higher level of compaction b. Silencing of gene transcription c. Localization at the periphery of the cell nucleus d. All of the above...
-
The fourth codon in an mRNA is GGG, which specifies glycine. Assuming that no amino acids are removed from the polypeptide, which of the following statements is correct? a. The third amino acid from...
-
Salen Company finances some of its current operations by assigning accounts receivable to a finance company. On July 1, 2017, it assigned, under guarantee, specific accounts amounting to $150,000....
-
What can scientists learn by comparing the fossilized skeletons of extinct primates with the bones of modern species?
-
The partnership of Sewell, Grange and Jones has just completed its first year in business. The partnership agreement stipulates that profits should be apportioned in the ratio of Sewell 3, Grange 2...
-
Peter and Paul, whose year end is 30 June, are in business as food wholesalers. Their partnership deed states that: a. profits and losses are to be shared equally; b. salaries are: Peter 20,000 per...
-
A, B, C and D were partners in a garage business comprising (1) petrol sales, (2) repairs and servicing and (3) second-hand car dealing. A was responsible for petrol sales, B for repairs and...
-
4. Methane burns in air with an equivalence ratio of =0.85, determine the composition of the products: What are the Mass Fractions of each species in the Products?
-
1. Describe the health and safety requirements of the area in which the installation activity is to take place, and the responsibility these requirements place on the learner 2. Describe the...
-
1. Which is easier for nucleation in solids, homogeneous or heterogeneous nucleation? What are the common defects present in solid solution (list at least five kinds of defects)? Are the defects...

Study smarter with the SolutionInn App


