Apply the appropriate material balance equations to verify that the lever rule gives the relative amount of
Question:
Apply the appropriate material balance equations to verify that the lever rule gives the relative amount of species in each phase along a tie line as depicted in Figure 8.2.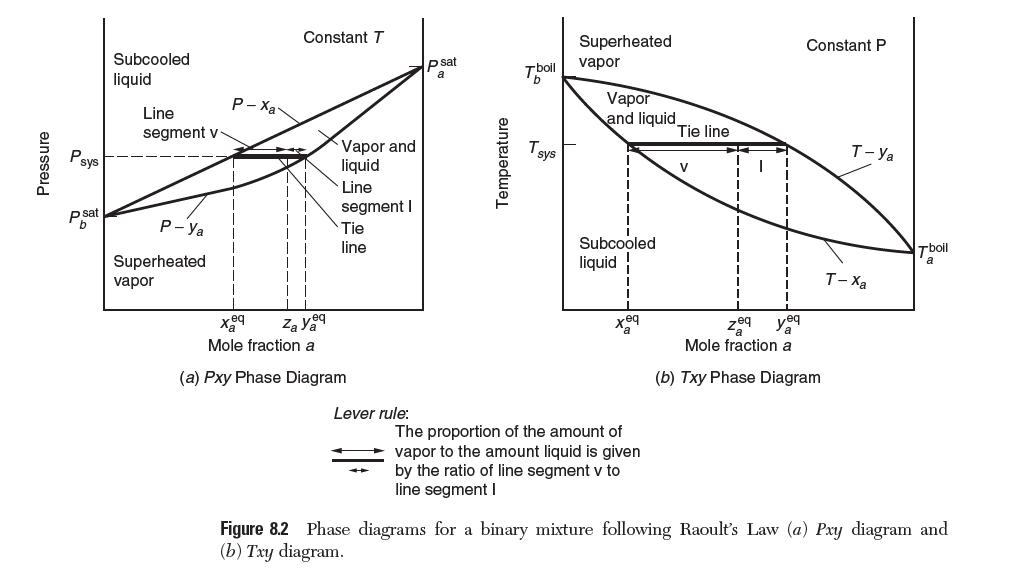
Transcribed Image Text:
Pressure Psys Psat Subcooled liquid Line segment v P-Ya Superheated vapor P-Xa Constant T Za yeq Vapor and liquid Line segment I Tie line Mole fraction a (a) Pxy Phase Diagram Lever rule: psat a Temperature Tboll T sys Superheated vapor Vapor and liquid Subcooled liquid eq The proportion of the amount of vapor to the amount liquid is given by the ratio of line segment v to line segment I Tie line V 1 Constant P Zeq yeq Mole fraction a (b) Txy Phase Diagram T-Ya T-Xa Tboil Figure 8.2 Phase diagrams for a binary mixture following Raoult's Law (a) Pxy diagram and (b) Txy diagram.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
A mass balance o...View the full answer

Answered By

John Aketch
I have a 10 years tutoring experience and I have helped thousands of students to accomplish their educational endeavors globally. What interests me most is when I see my students being succeeding in their classwork. I am confident that I will bring a great change to thins organization if granted the opportunity. Thanks
5.00+
8+ Reviews
18+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Engineering questions
-
Determine each of the following as being either true or false. If it is false, explain the reason why. 2 3-1 0 2 6-2 0 2
-
Planning is one of the most important management functions in any business. A front office managers first step in planning should involve determine the departments goals. Planning also includes...
-
Portray in words what transforms you would have to make to your execution to some degree (a) to accomplish this and remark on the benefits and detriments of this thought.You are approached to compose...
-
Why should one-time write-offs of fixed capital be used in absorption cost systems?
-
What is the reason of the participation rate in Alberta increased to 75 in 2008 then it decreased to 73.8 in 2010?
-
Let a curve be described by r = f(), where f() > 0 on its domain. Referring to the figure of Exercise 62, a curve is isogonal provided the angle is constant for all . a. Prove that is constant for...
-
The ages (in completed years) of the youngest leaders at the time of assuming office Find the mean, the median, and the mode of the data, if possible. If any measure cannot be found or does not...
-
What are the strength and direction of the electric field at the position indicated by the dot in FIGURE P23.37? Give your answer (a) In component form. (b) As a magnitude and angle measured cw or...
-
Consider a S corporation. The corporation earns $2.5 per share before taxes. The corporate tax rate is 35%, the tax rate on dividend income is 20%, and the personal income tax rate is set at 20%....
-
A binary vapor mixture contains 48% ethanol (a) in water (b) at 70C. Determine the pressure at which this vapor develops the fi rst drop of liquid. What is the liquid composition? The excess Gibbs...
-
A compressed liquid feed stream containing an equimolar mixture of n-pentane and n-hexane fl ows into a fl ash unit as shown in Figure E8.3 at fl ow rate F. At steady state, 33.3% of the feed stream...
-
What do you understand by existence of Fourier series?
-
Calculate the net present value (NPV) of a project with an initial investment of $50,000, expected cash flows of $10,000 per year for 5 years, and a discount rate of 12%. Show all your calculations...
-
Can you provide a overview of budgeting and forecasting in financial management? Describe the various types of budgets and forecasting methods. Additionally, explain how to prepare a budget and a...
-
Kyle has a new business opportunity, but his company needs to raise an additional $5,000,000 for the new project. Kyle wants to know the cost of this capital if he chooses to raise funds by selling...
-
In 2009, just four months after Najib Razak was elected Prime Minister of Malaysia, 1Malaysia Development Berhad (1MDB) was created. The wealth fund was established for development projects to better...
-
4. Let f be the function defined as follows: [lx-31 + 1, ax + bx, f(x) = {1x x < 3 x = 3 (a) If a = 3 and b = 2, is f continuous for all x? Justify your answer. (b) Describe all values of a and b for...
-
Think Like an Entrepreneur Perhaps you have fantasized about one day owning your own company, or maybe you have already started a business. Proposals are offers to a very specific audience whose...
-
On January 1, 2018, Khalid Ltd., which follows IAS 17, entered into an eight-year lease agreement for three dryers. Annual lease payments for the equipment are $28,500 at the beginning of each lease...
-
Show the conjugate bases of these species: a) H-0-H_bH-0 H CH-N-H d) H-C-C-H H
-
Complete these acid-base equations. Use the curved arrow method to show the electron movement in the reactions. Base Acid a) NH, + H: b) CHO + HO: Conjugate acid Conjugate base
-
Indicate whether each of these species is a Lewis acid, a Lewis base, or both: H 1. a) H-C I H d) CH3-N-H T H b) H-O-H :8-3-6: :ci: e) :CI-AI :CI: H c) H-B 1 H
-
The materiality calculation for Apollo Shoes seems appropriate, utilizing adjusted net income as the basis, adhering to a 5% threshold. This aligns with standard practices for profit-oriented...
-
At the end of its accounting period, December 31, of Year 1, Hsu's Financial Services has assets of $575,000 and stockholders' equity of $335,000. Using the accounting equation and considering each...
-
According to Alcntara Armando, it is a very important element to understand the direction that university systems are following, particularly in developing countries. Select one: The economic and...

Study smarter with the SolutionInn App


