Calculate the enthalpy of reaction at 298 K for the following reactions: (a) CH4(g) + 20(g) CO(g)
Question:
Calculate the enthalpy of reaction at 298 K for the following reactions: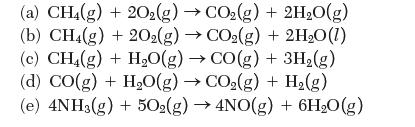
Transcribed Image Text:
(a) CH4(g) + 20(g) CO(g) + 2HO(g) (b) CH4(g) + 2O2(g) CO(g) + 2HO(1) (c) CH4(g) + HO(g) CO(g) + 3H(g) (d) CO(g) + HO(g) CO(g) + H(g) (e) 4NH3(g) + 502(g) 4NO(g) + 6H0(g)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (1 review)

Answered By

RAHUL SINGH SOUN
I have qualified gate, this makes me technically sound.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Engineering questions
-
Vehicle air bags protect passengers by using a chemical reaction that generates gas rapidly. Such a reaction must be both spontaneous and explosively fast. A common reaction is the decomposition of...
-
The Haber Bosch Process is a critically important chemical reaction run on an industrial scale. The reaction is shown below: 3 H+N2 =2 NH3 a) Using the thermodynamic tables in your textbook determine...
-
The molecule methylamine (CH3NH2) can act as a monodentate ligand. The following are equilibrium reactions and the thermochemical data at 298 K for reactions of methylamine and en with Cd2+ (aq); (a)...
-
Question 4 Tic-tac-toe (also known as noughts and crosses) is a game for two players, X and O, who take turns marking the spaces in a 3x3 grid. The player who succeeds in placing three of their marks...
-
Assume Mess stock has a beta of 1.2. If the risk-free rate is 7 percent, and the market return is 10 percent, what is the expected return on Mess stock?
-
What will the monthly payment be if you borrow $30,000 for 36 months through Toyota at 0.99% compounded monthly? (We will use monthly compounding for simplicity.)
-
Describe a variety of negligent errors that have application to all healthcare professionals.
-
Professor Schmidt, a geologist, has agreed to purchase McDonalds farm for a price of $2,000 per acre, which corresponds to the price of good quality farmland in the vicinity. However, Schmidt, on the...
-
Compute the standard deviation for your two asset portfolio Asset X: Weight = 0.5 Standard deviation = 0.0595 Correlation = 0.3568 Asset Y: Weight = 0.5 Standard deviation = 0.0930 Correlation =...
-
Calculate the adiabatic fl ame temperature of acetylene gas at a pressure of 1 bar under the following conditions. The reactants are initially at 298 K. Assume that the acetylene reacts completely to...
-
One mole of saturated liquid propane and 1 mole of saturated vapor are contained in a rigid container at 0C and 4.68 bar. How much heat must be supplied to evaporate all of the propane. At 0C, You...
-
The KMP Metal Machining Company produces widgets according to customer order. The company has determined that widgets can be produced on three different machine tools: M1, M2, or M3. An analysis of...
-
1. Describe FIVE (5) roles of cruise service to the growth of Malaysia, Singapore and Thailand economy. (10 marks)
-
Design a supply chain diagram of how BMW integrates blockchain?
-
(4 points) In the circuit shown, find all current 11,12, and 13. e Kirchhoff law) 13 1 1 2002 www www 5 27 V 11 V www 9
-
With the use of examples of the types of questions that are asked in determining a sample, as well as at least five principles that samples must adhere to , discuss the concept of sampling.
-
In the figure, the particles have charges 91 = -92 = 290 nC and 93 = -94 - 98 nC, and distance a = 6.8 cm. What are the (a) x and (b) y components of the net electrostatic force on particle 3? (a)...
-
An analyst predicted last year that the stock of Logistics, Inc., would offer a total return of at least 10 % in the coming year. At the beginning of the year, the firm had a stock market value of...
-
I frequently use NY Times and CNN and am aware of Fox News but I never use it. I visit these sites, NY Times and CNN, a few times a week whenever I have to research something or see something on...
-
Uscharidin is the common name of a poisonous natural product having the structure shown. Locate all of the following in uscharidin:
-
Write the structural formula of a compound of molecular formula C4H8Cl2 in which (a) All the carbons belong to methylene groups (b) None of the carbons belong to methylene groups
-
Female tiger moths signify their presence to male moths by giving off a sex attractant. The sex attractant has been isolated and found to be a 2-methyl-branched alkane having a molecular weight of...
-
Calculating USLE (#3-5): Assume maximum soil loss of 5 tons/acre all questions. Use the tabular data to help answer the questions. C-factors are split into Tables 4A and 4B therefore C-factor = 4A 4B...
-
Suppose that adidas collaborated with the NFL to create a series of sports shoes and apparel incorporating the colors and logos of the various NFL teams, and bearing both the adidas and NFL brands....
-
Pizza Hut had free cash flows (FCF) of $1.05mm this past year. Projected FCFs for the next five years are $1.07mm, $1.10mm, $1.15mm, $1.20mm, and $1.25mm, respectively. After five year FCFs will be...

Study smarter with the SolutionInn App


