Calculate the second virial coeffi cient, B, for CH4 over the temperature range 100900 directly from the
Question:
Calculate the second virial coeffi cient, B, for CH4 over the temperature range 100–900 directly from the Lennard-Jones parameters. Compare the values to the following data reported in the literature.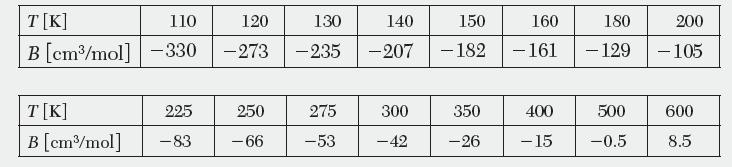
Transcribed Image Text:
T[K] B [cm/mol] T[K] B [cm/mol] 110 -330 225 -83 120 130 -273 - 235 250 -66 275 -53 140 150 160 180 -207 -182 - 161 - 129 300 -42 350 -26 400 -15 500 -0.5 200 -105 600 8.5
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
We can solve this problem by substituting the LennardJones potential Equation 410 into the molecular ...View the full answer

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Engineering questions
-
In this problem we seek to develop an expression for the van der Waals constants a and b in terms of molecular parameters using the Sutherland model for potential energy. (a) Show that writing the...
-
The square-well potential function is given by: Answer the following questions: (a) Sketch a plot of square-well potential energy versus distance. (b) Using this potential function, develop an...
-
The second virial coeffi cient for argon is reported versus temperature in the following table. Explain the trend with temperature in terms of dominant intermolecular interactions. What can you say...
-
ABC Pty Ltd would like to set up a Virtualisation Platform on their organisation. You have been hired by Company to be their network and system administrator to implement virtualisation for...
-
What is the IAS Regulation?
-
All-Star Automotive Company experienced the following accounting events during Year 1: 1. Performed services for $25,000 cash. 2. Purchased land for $6,000 cash. 3. Hired an accountant to keep the...
-
For the standpipe system shown in Fig. P8.94, calculate the flow rate for \(H=4.0 \mathrm{ft}, D=6.77\) in., \(d=0.125\) in., and \(L=48\) in. The fluid is \(70^{\circ} \mathrm{F}\) water. Assume...
-
At December 31, balances in Manufacturing Overhead are: Shimeca Companydebit $1,200, Garcia Companycredit $900. Prepare the adjusting entry for each company at December 31, assuming the adjustment is...
-
c) Please analyse figure below: i. ii. page: ABBACDEFGCDF Frame: 1 A A FL 2 BB 131 E 3 C G 4 D C Figure 2 D F What is the replacement policy that been used in Figure 2. Please support your answer...
-
Determine the molar volume of copper at 500C from the data in Table 4.4. TABLE 4.4 Molar Volume, Thermal Expansion Coefficient, and Isothermal Compressibility of Some Liquid and Solid Species at 20C...
-
Calculate an expression for the second virial coeffi cient, B, for the hard sphere potential model.
-
What is the angular momentum of a 0.270-kg ball revolving on the end of a thin string in a circle of radius 1.35 m at an angular speed of 10.4 rad/s?
-
Determine the net working capital funding needs, given the following: Cash = $15,000 Inventory = $30,000 Average Receivables = $20,000 Average Payables = $25,000 2. Determine the seasonal funding...
-
How do shifts in consumer preferences impact profit margins, and what adaptive measures can companies adopt to mitigate associated risks ?
-
An optimal combination of decisions relating to investment, financing and dividends will maximise the value of the firm to its shareholders, Examine? What role should the finance manager play in the...
-
A Lease Go to an apartment complex office and ask for a copy of a lease. Bring the lease to class and read the contents. Then make one list of the renters responsibilities and another list of the...
-
The Assembly Department of Interface, Inc., manufacturer of computers, had 500 units of beginning inventory in September, and 4,000 units were transferred to it from the Production Department. The...
-
Trost Leasing Company buys equipment for leasing to various manufacturing companies. On October 1, 2010, Trost leases a press to Shumway Shoe Company. The cost of the machine to Trost was $196,110,...
-
A consultant is beginning work on three projects. The expected profits from these projects are $50,000, $72,000, and $40,000. The associated standard deviations are $10,000, $12,000, and $9,000....
-
Suffocation of naphthalene is reversible at elevated temperature. A different isomer of naphthalenesulfonic acid is the major product at 160C than is the case at 0C. Which isomer is the product of...
-
On being heated with sulfur trioxide in sulfuric acid, 1, 2, 4, 5-tetramethylbenzene was converted to a product of molecular formula C10H14O3S in 94% yield. Suggest a reasonable structure for this...
-
When benzene is prepared from coal tar, it is contaminated with thiophene, from which it cannot be separated by distillation because of very similar boiling points. Shaking a mixture of benzene and...
-
Suppose that a consumer has preferences defined over the consumption set R2 = {(x1, x2) x = [0, 0), x2 = [0, )}. In other words, this consumer has preferences defined over the set of all bundles...
-
Should governments regulate markets to prevent high concentration ratios? Explain and provide reference.
-
2. Suppose there are n identical consumers, each with a utility function u(x, y) = ax = x+y. Each consumer has different disposable income w. The price of good x is p dollars, and the price of...

Study smarter with the SolutionInn App


