Consider a mixture of 1-propanol (a) and water (b). At 25C, the three-suffi x Margules equation parameters
Question:
Consider a mixture of 1-propanol
(a) and water (b). At 25°C, the three-suffi x Margules equation parameters are: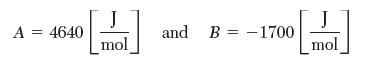
Does this binary mixture form an azeotrope at 25°C? If so, at what pressure does the azeotrope form?
Transcribed Image Text:
A = 4640 J mol and B-1700 J mol
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)

Answered By

Krishnavendra Y
I am a self motivated financial professional knowledgeable in; preparation of financial reports, reconciling and managing accounts, maintaining cash flows, budgets, among other financial reports. I possess strong analytical skills with high attention to detail and accuracy. I am able to act quickly and effectively when dealing with challenging situations. I have the ability to form positive relationships with colleagues and I believe that team work is great key to performance. I always deliver quality, detailed, original (0% plagiarism), well-researched and critically analyzed papers.
5.00+
4+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Engineering questions
-
a. Example 13-1: Batch Reactor with an Exothermic Reaction Wolfram 1. Adiabatic Case: Use Wolfram to see whether you can find a trajectory that is ready to ignite and whose trajectory looks like a...
-
Let r and s be solutions to the quadratic equation x 2 b x + c = 0. For n N, define d0 = 0 d1 = r s dn = b dn1 c dn2 (n 2) Prove that dn = r n s n for all n N. [4 marks] (b) Recall that a commutative...
-
(a) LEP Table 12-2: Exothermic Reaction with Heat Exchange Download the Polymath, MATLAB, Python, or Wolfram codes for the algorithm and data given in Table T12-2 for the exothermic gas phase...
-
In Exercises 5 and 6, the transition matrix P for a Markov chain with states 0 and 1 is given. Assume that in each case the chain starts in state 0 at time n = 0. Find the probability that the chain...
-
June Devlin operates a Water Bottling Service in Yellowknife. All transactions requiring the payment of cash are recorded in the cash payments journal (page 14). The account balances as of May 1,...
-
Nicky Pope filed a false imprisonment lawsuit against the Rostraver Shop n Save store (Shop n Save) and its manager, Howard Russell, on the basis of the incident described below. Pope and Russell...
-
Verify the three predictions (i.e., Equations 3.27, 3.40, and 6.67) for the Young's modulus of the glass microsphere-reinforced polyester composite in Figure 6.42 for the specific case of a particle...
-
Equipment acquired on January 6, 2011, at a cost of $714,000, has an estimated useful life of 12 years and an estimated residual value of $44,400. a. What was the annual amount of depreciation for...
-
Shankar Company uses a perpetual system to record inventory transactions. The company purchases inventory on account on February 2 for $35,000 and then sells this inventory on account on March 17 for...
-
At 20C and 0.073 bar, a binary liquid mixture of cyclohexane (1) and toluene (2) is in vapor liquid equilibrium. The liquid mole fraction of cyclohexane is measured to be x1 = 0.471. Assume that the...
-
A binary mixture of water (1) and benzene (2) is in vaporliquid equilibrium at a liquid mole fraction x1 = 0.6 and a pressure of 74.5 kPa. The liquid phase nonideality can be described by the...
-
Solve. Find exact solutions. x + 4 = x - 4 = 2 -
-
What is your analysis of Aviva Investors' campaign against Vedanta? What are the benefits to Aviva?
-
Assume the interest rate is 10% and quarterly compounding. 1. You are going to receive $10,000 in five years. What is the present value? 2. You are going to receive $2,500 at the end of each quarter...
-
Discuss the maSince the late 1980's many national financial markets got aboard the liberalization process to enhance global integration of all the financial markets. Required: i) Discuss the...
-
How do terrorist organizations typically convert cryptocurrencies into fiat currency, considering the challenges associated with direct conversion on traditional exchanges?
-
Porsche hedges by buying two-year European at-the-money put options on USD (providing to Porsche the right to sell USD, receiving Euros, at the strike exchange rate) in sufficient quantity to have...
-
Name and briefly describe the five product mix pricing decisions.
-
9.Consider the reaction 3NO2(g)+H2O=2HNO3(aq)+NO(g) where Delta H=-137 kJ.How many kilojoules are released when 92.3g of NO2 reacts?
-
Calculate the final pressure of a sample of water vapour that expands reversibly and adiabatically from 87.3 Torr and 500 dm3 to a final volume of 3.0 dm3. Take Y = 1.3
-
When 178J of energy is supplied as heat to 1.9 mol of gas molecules, the temperature of the sample increases by 1.78 K. Calculate the molar heat capacities at constant volume and constant pressure of...
-
When 2.0 mol CO2 is heated at a constant pressure of 1.25 atm, its temperature increases from 250 K to 277 K. Given that the molar heat capacity of CO2 at constant pressure is 37.11 J K-1 mol-1,...
-
Read the NAEYC Administrators Code of Ethical Conductand answer the following questions. Part 2 Questions What is the purpose of the administrator supplement of the NAEYC Code of ethical conduct?...
-
The Stockholder s Equity accounts of a corporation on January 1 , 2 0 2 2 , were as follows: Preferred Stock ( 5 % , $ 1 0 par, cumulative, 5 , 0 0 0 shares authorized ) $ 2 8 , 0 0 0 Common Stock...
-
Ronny and Assoc. issued a check for $2,400 to pay salaries. This transaction would be recorded with an increase on the left-hand side of which type of account ?

Study smarter with the SolutionInn App


