Consider a system containing water in the following states. What phases are present? (a) P = 10
Question:
Consider a system containing water in the following states. What phases are present?
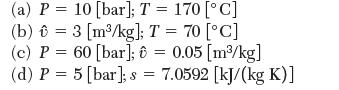
Transcribed Image Text:
(a) P = 10 [bar]; T = 170 [C] (b) = 3 [m/kg]; T = 70 [C] (c) P = 60 [bar]; 6 = 0.05 [m/kg] (d) P = 5 [bar]; s = 7.0592 [kJ/(kg K)]
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (3 reviews)
The provided image contains thermodynamic properties of water at different ...View the full answer

Answered By

Ehsan Mahmood
I’ve earned Masters Degree in Business Studies and specialized in Accounts & Finance. Couple with this, I have earned BS Sociology from renowned institute of Pakistan. Moreover, I have humongous teaching experience at Graduate and Post-graduate level to Business and humanities students along with more than 7 years of teaching experience to my foreign students Online. I’m also professional writer and write for numerous academic journals pertaining to educational institutes periodically.
4.90+
248+ Reviews
287+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Engineering questions
-
Journalise the June transactions. (Note: Enter debit entries first, followed by credit entries in the general journal. Credit account titles are automatically indented when the amount is entered - do...
-
answer the question clearly You are building a flight-control system for which a convincing safety case must be made. Would you assign the tasks of safety requirements engineering, test case...
-
In this question you will be asked to reflect on a project you have been involved in or observed, in which a design evolved, or could have evolved, through applying a theory of user behaviour. You...
-
Rafael Espinal's leadership approach is best categorized as what type of leadership? spiritual adaptive authentic servant
-
BNC, a closely held corporation, was organized in 1987. To date, it has accumulated more than $10 million after-tax income. This year, BNC's taxable income is $750,000, and its federal tax is...
-
A rock group needs to choose 3 songs to play at the annual Battle of the Bands. How many ways can they choose their set if have 15 songs to pick from?
-
We continue the analysis begun in Exercise 1.7 by examining the relation between y = LIFEEXP and x = FERTILITY, shown in Figure 2.12. Fit a linear regression model of LIFEEXP using the explanatory...
-
On November 1, 2007, Columbo Company adopted a stock option plan that granted options to key executives to purchase 30,000 shares of the companys $10 par value common stock. The options were granted...
-
Project Overview: The project is used to illustrate the process and documentation required for a network security design. This project presents a scenario in which the RGIT SOFTWARE AUSTRALIA has...
-
Estimate the speed at which the average oxygen molecule is moving in the room that you are in.
-
The ideal gas model is one example of an equation of state. Why do you think it is termed an equation of state?
-
A regional express delivery service company recently conducted a study to investigate the relationship between the cost of shipping a package (Y), the package weight (X1), and the distance shipped...
-
Read the National Association for the Education of Young Children (NAEYC) 2003 position statement on early childhood curriculum, assessment, and program evaluation, available at...
-
answer the following questions with the data provided. Show calculations by illustrating using the funnel model. Sales = 90 units/month (In flow) Production = 90 units/month (outflow) Backlog = 225...
-
Harry says great leaders are born that way. Sally says great leaders are not born great, they are "made" (through education, experience, hard work, etc.). What is your position on this? Regardless of...
-
Part One Prepare your own lessons learned report (1-2 pages, single-spaced) based on personal reflections of what you learned from this class, including the team projects and presentations. Do not...
-
Motivation - What motivates you to complete this course? Possible answers: Parents - if not in college then out you go (FEAR). College degree opens doors to jobs (WANTS, MONEY, Wealth). College is...
-
Betty Melton does not understand why the overhead volume variance indicates that fixed overhead costs are under-or-overapplied. Clarify this matter for Betty.
-
Consider model (9.18). What is the effect on the model parameter estimates, their standard errors, and the goodness-of-fit statistics when (a) The times at risk are doubled, but the numbers of deaths...
-
Treatment of 3-methyl-2-butanol with hydrogen chloride yielded only a trace of 2-chloro-3-methylbutane. An isomeric chloride was isolated in 97% yield. Suggest a reasonable structure for this product.
-
Write the structure of the principal organic product to be expected from the reaction of 1-bromopropane with each of the following: (a) Sodium iodide in acetone (CH3CONa) (b) Sodium acetate in acetic...
-
All the reactions of 1-bromopropane in the preceding problem give the product of nucleophilic substitution in high yield. High yields of substitution products are also obtained in all but one of the...
-
There are many reasons why the budget process may be compromised. This has a greater potential impact of programs funded by the discretionary budget (which is determined yearly) as compared to...
-
You are required to provide a written report with evidence attached that proves you have identified and evaluated the food preferences of customer groups with differing characteristics so you can use...
-
According to the text, which method of asset valuation generally excludes intangible assets?

Study smarter with the SolutionInn App


