Consider water at 250C and 800 kPa. Determine a value for the partial derivative by the following
Question:
Consider water at 250°C and 800 kPa. Determine a value for the partial derivative 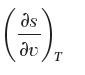
by the following methods:
(a) Use the Redlich–Kwong equation of state.
(b) Use the steam tables directly.
(c) Use the steam tables together with the Maxwell relation given in Equation (5.18).
Compare your answers and explain which one you think is the most accurate.
Transcribed Image Text:
s 2) T
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)

Answered By

Akshay Agarwal
I am a Post-Graduate with a specialization in Finance. I have been working in the Consulting industry for the past 8 years with a focus on the Corporate and Investment Banking domain. Additionally, I have been involved in supporting student across the globe in their academic assignments and always strive to provide high quality support in a timely manner. My notable achievements in the academic field includes serving more than 10,000 clients across geographies on various courses including Accountancy, Finance, Management among other subjects. I always strive to serve my clients in the best possible way ensuring high quality and well explained solutions, which ensures high grades for the students along-with ensuring complete understanding of the subject matter for them. Further, I also believe in making myself available to the students for any follow-ups and ensures complete support and cooperation throughout the project cycle. My passion in the academic field coupled with my educational qualification and industry experience has proved to be instrumental in my success and has helped me stand out of the rest. Looking forward to have a fruitful experience and a cordial working relationship.
5.00+
179+ Reviews
294+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Engineering questions
-
Determine Z and V for steam at 250 C and 1.800 kPa by the following: (a) The truncated virial equation [Eq. (3.40)] with the following experimental values of virial coefficients: (b) The truncated...
-
Let A, B be sets. Define: (a) the Cartesian product (A B) (b) the set of relations R between A and B (c) the identity relation A on the set A [3 marks] Suppose S, T are relations between A and B, and...
-
A mixture of 60% ethylene and 40% acetylene by moles is at 6 MPa, 300 K. The mixture flows through a preheater where it is heated to 400 K at constant P. Using the Redlich Kwong equation of state...
-
A curve C in three dimensions is given parametrically by (x(t), y(t), z(t)), where t is a real parameter, with a t b. Show that the equation of the tangent line at a point P on this curve where t...
-
1- What are the most important project closure activities? How can you ensure the quality of the project closure process? 2- As a project manager, you may be asked to facilitate lessons learned as...
-
Determine the normalized specifications for the analog, lowpass, Chebysev filter corresponding to the highpass filter: \[\begin{aligned} A_{\mathrm{p}} & =0.2 \mathrm{~dB} \\ A_{\mathrm{r}} & =50...
-
Imagine you are working for a company that sells teleconferencing equipment. Draft a value proposition for selling your equipment to a sales manager who has ten salespeople traveling two weeks a...
-
Kershaw Electric sold $6,000,000, 10%, 10-year bonds on January 1, 2020. The bonds were dated January 1, 2020, and paid interest on January 1. The bonds were sold at 98. Instructions a. Prepare the...
-
Briefly discuss the differences between the DCF method, the DDM method, and the market multiples method of valuing a company. What are the strengths AND weaknesses to each method? If you were asked...
-
A gas can be described by the following equation of state: where b = 5 * 10 24 m3/mol and c P ideal gas = 35 J/ 1mol K2 . (a) Develop an expression of creal P for this gas. (b) One mole of this gas...
-
Your company has just developed a new refrigeration process. This process uses a secret gas, called Gas A. You are told that you need to come up with thermodynamic property data for this gas. The...
-
During October, All Pets Veterinary Clinic Ltd. completed the following transactions: Oct. 1 Dr. Squires deposited $8,000 cash in the business bank account. The business issued common shares to her....
-
What other product issues can influence a companys product or brand reputation in a market abroad?
-
The financial statements for the governmental funds use the a. Economic resources measurement focus and the accrual basis of accounting. b. Current financial resources measurement focus and the...
-
For the year ended December 31, 20X3, how much should Dodge report as net patient service revenue? a. $4,500,000 b. $5,000,000 c. $5,500,000 d. $5,740,000 Under its established rate structure, Dodge...
-
What are the five ways that a company can blend its product and promotional strategies?
-
Which of the following funds can be major assuming they meet the appropriate tests? a. The parking meter special revenue fund and the water utility enterprise fund. b. The fire station capital...
-
Must an agency fund be used to account for withholding taxes, retirement contributions, and (if applicable) social security taxes of General Fund employees? Explain.
-
The Dow Jones Industrial Average reached a high of $ 7801.63 on December 29, 1997. Recall from Example 18.4 that it reached a high of $ 1003 on November 14, 1972. The Consumer Price Index for...
-
At an altitude of 15 km the temperature is 217 K and the pressure 12.1 kPa. What is the mean free path of N, molecules? (a= 0.43 NM2)
-
How many collisions per second does an N2 molecule make at an altitude of 15 km? (See Exercise 21.4b for data.)
-
Calculate the mean free path of carbon dioxide molecules using a= 0.52 nm at 25C and (a) 15 atm, (b) 1.0 bar, (c) 1.0 Torr.
-
A five-year regular annuity has a present value of $1,000. If the interest rate is 8 percent, which answer is closest to the annuity payment?Group of answer choices$181.62$231.91$184.08$250.44
-
2. Perform the following tasks: a) Consider a portfolio of three funds, namely funds 1, 2 and 3, with weights W, W and W3, respectively. Construct the portfolio variance. [4 marks] b) Construct in...
-
$200,000 U.S. Treasury 7 7/8% bond maturing in 2002 purchased and then settled on October 23, 1992, at a dollar price of 105-20 (this is the clean price) with a yield to maturity of 7.083% with the...

Study smarter with the SolutionInn App


