Determine the change in entropy of an ideal gas with constant heat capacity, cP = (7/2)R, between
Question:
Determine the change in entropy of an ideal gas with constant heat capacity, cP = (7/2)R, between the following states: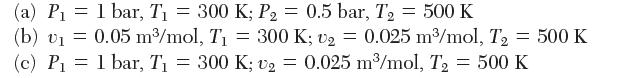
Transcribed Image Text:
(a) P = 1 bar, T = 300 K; P = 0.5 bar, T = 500 K (b) v = 0.05 m/mol, T = 300 K; v = 0.025 m/mol, T (c) P = 1 bar, T = 300 K; = 0.025 m/mol, T = 500 K T2 = 500 K
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 33% (3 reviews)

Answered By

Allan Olal
I have vast tutoring experience of more than 8 years and my primary objective as a tutor is to ensure that a student achieves their academic goals.
4.70+
78+ Reviews
411+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Engineering questions
-
Euler's original article about the Konigsberg Bridge Problem, which is dated 1736, presents a second similar problem with two islands, four rivers flowing around them, and 15 bridges connecting...
-
Problem 3.27 consists of an irreversible process in which an ideal gas with constant heat capacity was compressed in a pistoncylinder assembly. As part of this problem, you were asked to calculate...
-
An ideal gas with constant heat capacities undergoes a change of state from conditions T1, P1 to conditions T2, P2. Determine (H (J mol-1) and (S (J mol-1 K-1) for one of the following cases. (a) T1...
-
What other advice can you offer owners of seasonal businesses about coping with the effects of their companies highly irregular sales patterns? About managing cash flow in general?
-
Norris, Inc. is preparing its budget. Production for May will be 500 widgets, then 600 for June, 700 for July, and 800 for August. Each widget requires 4 pounds of plastic and each pound costs $1.00....
-
Brilliant Equipment purchased machinery from a Japanese firm and must make a payment of 313.25 million in 45 days. The bank quotes a forward rate of 103.01/$ to buy the required yen. What is the cost...
-
The smooth, concrete-lined channel shown in Fig. P10.46 is built on a slope of \(2 \mathrm{~m} / \mathrm{km}\). Determine the flowrate if the depth is \(y=1.5 \mathrm{~m}\). Figure P10.46 -6 m- 1.0 m...
-
As the accountant for Veneskey & Sons, you have been hired to prepare the payroll and everything that goes along with it for OlFashion Industries which has 4 employees. Their necessary payroll...
-
Throughout the course, have learned about various concepts in international finance. This assignment provides an opportunity for you to apply those concepts and reflect on important topics in...
-
Compare the change in entropy (a) when water is heated from its freezing point to its boiling point at 1 atm and (b) when saturated liquid water is vaporized at 1 atm.
-
Calculate the change in entropy of the universe for the process described in Problem 2.28. Repeat for Problem 2.29. Problem 2.28 Consider a pistoncylinder assembly that contains 1 mole of ideal gas,...
-
Leadership is one of the holy grails of management and organisational behaviour. however complex or nebulous the subject area may appear, an understanding of leadership is an essential part of your...
-
KUALA LUMPUR: The FBM KLCI hastened its decline headed into midday as the blue-chip counters fell in unison on the Budget 2022 proposal of a one-off special tax on high-income companies. At 12.30pm,...
-
Discuss how a company can improve its production and sales
-
Assume that you and some of your classmates will form a partnership of some type for the sole purpose of investing in real estate. You meet at your favorite restaurant and draft an informal agreement...
-
Draw a sketch of Graph A from this lesson and carefully circle when a hand was in contact with the The sketch only must show the shape of the graph. It does not have to be exact. Mark an at the point...
-
How does public back assume a critical part in a money contextual analysis, and what are the vital contemplations and difficulties legislatures and policymakers face while overseeing public assets...
-
If an operating lease requires the payment of uneven rental amounts over its life, how should the lessee recognize rental expense?
-
As indicated by mutual fund flows, investors tend to beat the market seek safety invest in last year's winner invest in last years loser
-
Does sodium borohydride reduction of D-ribose yield an optically active product? Explain.
-
Which of the following would be expected to give a positive test with Benedicts reagent? Why? (a) D-Galactitol (see structure in margin) (d) D-Fructose (b) L-Arabinose (e) Lactose (c) 1,...
-
Another hexose gives the same aldaric acid on oxidation as does D-glucose. Which one?
-
Assume that the government is unable to change the amount of public debt that the private sector holds. Consider both fixed and flexible exchange rate regimes. Explain how would the nominal and real...
-
The Welton Mfg Company of Sioux Falls has a balance sheet with the following capital structure: Common stock, Preferred stock and Debt. Quantity 250,000 shares price $18 par value 105% of par value...
-
An individual has the genotype AO, hh. What antigens would be present on the red cells of this individual? What antibodies does this individual have?

Study smarter with the SolutionInn App


