Evaluate the derivative: for a pure species that follows the PengRobinson equation of state. The subscript T,P
Question:
Evaluate the derivative: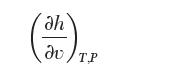
for a pure species that follows the Peng–Robinson equation of state. The subscript T,P indicates that both temperature and pressure are held constant.
Transcribed Image Text:
dh v T.P
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (3 reviews)

Answered By

Lamya S
Highly creative, resourceful and dedicated High School Teacher with a good fluency in English (IELTS- 7.5 band scorer) and an excellent record of successful classroom presentations.
I have more than 2 years experience in tutoring students especially by using my note making strategies.
Especially adept at teaching methods of business functions and management through a positive, and flexible teaching style with the willingness to work beyond the call of duty.
Committed to ongoing professional development and spreading the knowledge within myself to the blooming ones to make them fly with a colorful wing of future.
I do always believe that more than being a teacher who teaches students subjects,...i rather want to be a teacher who wants to teach students how to love learning..
Subjects i handle :
Business studies
Management studies
Operations Management
Organisational Behaviour
Change Management
Research Methodology
Strategy Management
Economics
Human Resource Management
Performance Management
Training
International Business
Business Ethics
Business Communication
Things you can expect from me :
- A clear cut answer
- A detailed conceptual way of explanation
- Simplified answer form of complex topics
- Diagrams and examples filled answers
4.90+
46+ Reviews
54+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Engineering questions
-
(a) LEP Table 12-2: Exothermic Reaction with Heat Exchange Download the Polymath, MATLAB, Python, or Wolfram codes for the algorithm and data given in Table T12-2 for the exothermic gas phase...
-
a. Example 13-1: Batch Reactor with an Exothermic Reaction Wolfram 1. Adiabatic Case: Use Wolfram to see whether you can find a trajectory that is ready to ignite and whose trajectory looks like a...
-
The following variables are used in an implementation of the algorithm: ar is the count of active readers rr is the count of reading readers aw is the count of active writers ww is the count of...
-
Marketing efforts to produce, promote, and reclaim environmentally sensitive products are called ________________ .
-
You need to examine approaches used by the three components of the criminal justice system - law enforcement, courts, and corrections - for the prevention of crime. Include the following in your...
-
Design a bandpass filter satisfying the specification below using the Hamming, Hann, and Blackman windows: \[\begin{aligned}M & =10 \\\Omega_{\mathrm{c}_{1}} & =1.125 \mathrm{rad} /...
-
Review your answers to the previous two questions. Comment on the persuasiveness of evidence from each test. Explain any factors that would affect your assessment. Fellowes and Associates Chartered...
-
Multiple Choice Questions 1. Which one of the following managerial accounting approaches attempts to allocate manufacturing overhead in a more meaningful fashion? a. Activity-based costing b....
-
labour cost. Last year, the company sold 30,000 of these balls, with the following results: Sales (30,000 balls) $750,000 Variable expenses Contribution margin 450,000 300,000 Fixed expenses...
-
Using the van der Waals equation, fi nd an expression for the derivative: in terms of a, b, cP, R, v, and T. ( ) . dh . 50
-
Show that an ideal gas follows the cyclic relationship in P, v, and T.
-
The taxpayer is an antiques collector and is going to sell an antique purchased many years ago for a large gain. The facts and circumstances indicate that the taxpayer might be classified as a dealer...
-
Determine the annual net cost of these checking accounts: a. Monthly fee $5, check-processing fee of 25 cents, average of 19 checks written per month b. Annual interest of 2.5% paid if balance...
-
Which of the following accounts of a governmental unit is credited when taxpayers are billed for property taxes? a. Estimated Revenue. b. Revenue. c. Appropriations. d. Fund BalanceAssigned for...
-
How is the composition of FDI sources changing with respect to developed and developing nations?
-
Why might 100 percent ownership of a subsidiary not give a company complete control?
-
Which of the following situations will increase a governmental units fund balance at the fiscal year-end? a. Appropriations are less than expenditures and budgetary fund balance assigned for...
-
Central Duplicating Internal Service Fund. As of September 30, 2010, the Central Duplicating Fund of the Town of Fredericksburg had the following post-closing trial balance: During the fiscal year...
-
Provide a few individual examples who revealed what aspects of emotional intelligence?
-
A Knudsen cell was used to determine the vapour pressure of germanium at 1000C. During an interval of7200 s the mass loss through a hole of radius 0.50 mm amounted to 43 ug, what is the vapour...
-
An atomic beam is designed to function with (a) Cadmium, (b) Mercury. The source is an oven maintained at 380 K, there being a small slit of dimensions 1.0 cm x 1.0 x 10-3 cm. The vapour pressure of...
-
The resistances of a series of aqueous NaCI solutions, formed by successive dilution of a sample, were measured in a cell with cell constant (the constant C in the relation K= C/R) equal to 0.2063...
-
Today is January 1, 2022. You plan to retire on January1, 2052. Starting today, you want to deposit a fixed amount every year into the All-Smiles Retirement Fund which provides an expected annual...
-
Suppose you receive a 4-year ordinary annuity of $1,000 per year and deposit the money in a savings account at the end of each year. The account earns interest at a rate of 6 percent compounded...
-
Use Ito's lemma to show that (a) sdW(s) =tW (t) - W(s)ds (b) W(s)dW(s) = W (t) fW(s)ds (c) E(W(t)) = 31, -

Study smarter with the SolutionInn App


