The Gibbs energy of mixing, gmix in [J/mol], of a binary liquid mixture of water (1) and
Question:
The Gibbs energy of mixing, Δgmix in [J/mol], of a binary liquid mixture of water (1) and 1-propanol (2) vs. mole fraction water (x1)is shown below. These data are at a temperature at 40°C.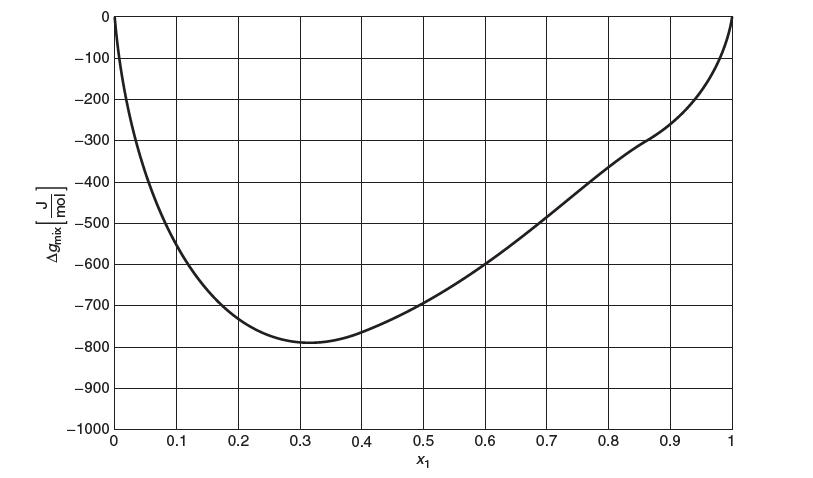

Transcribed Image Text:
A9mix mol! 0 -100 -200 -300 -400 -500 -600 -700 -800 -900 -1000 0 0.1 0.2 0.3 0.4 0.5 X1 0.6 0.7 0.8 0.9
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)

Answered By

Alakhdeep Singh
I am teaching in chegg site and I am taking home tuition also
Till now I have teached above 100 students
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Engineering questions
-
How long will it take will it take a 1500 kW motor to lift a 400 kg piano to a sixt-storey window 20 m above the ground?
-
Determine the slope and displacement at point C. EI is constant. Assume A is a pin. Use the method of virtual work. A 2 k/ft 12 ft B Probs. 8-30/31 6 ft C
-
The excess Gibbs energy of solutions of methylcyclohexane (MCH) and tetrahydrofuran (THF) at 303.15 K was found to fit the expression GE=RTx (1- x) {0.4857 - 0.1077(2x -1) + 0.0191(2x- 1)2} where x...
-
Based on Exhibits 5 and 6, the value of the lower one-period forward rate is closest to: A. 3.5122%. B. 3.5400%. C. 4.8037%. Meredith Alvarez is a junior fixed-income analyst with Canzim Asset...
-
Review the case study "Alaska Airlines: Navigating Change" and then complete the following: https://www.kotterinternational.com/8-steps-process-for-leading-change/ A- State what actually occurred in...
-
Derive the two-dimensional matrix representation Tic)=(2) Tin)=(3) Tex)=(37) (69) T(c2b)= 1 TO)-(71) 10-(11) TO=(9) = for the group D3, using the basis (e1, e2) defined in the following figure.
-
In each of the following situations, formulate the null and alternative hypotheses. a. A new product will be introduced if it is preferred by more than 70 percent of the consumers in the target...
-
Select an AIS system segment (such as purchasing, sales, or payroll), investigate, and write a report on the tangible and intangible costs and benefits associated with your selection.
-
flower pot sitting on a table. Sign Convention: Force Diagram: Net Force Equation(s): , Subscript Definitions: A chandelier hanging from a chain. Sign Convention: Force Diagram: Net Force...
-
A binary mixture of species a and b behaves as an ideal gas at 300 K and 1 bar. Calculate the partial molar Gibbs energy of species a, Ga, and the total solution Gibbs energy, g, at the following...
-
A group of students came across an unsuspected supply of laboratory alcohol containing 96 weight % ethanol (EtOH) and 4 weight % water (H2O) . As an experiment, they wanted to see ifthey could make...
-
A compound of formula C 6 H 10 O 2 shows only two absorptions in the proton NMR: a singlet at 2.67 ppm and a singlet at 2.15 ppm. These absorptions have areas in the ratio 2:3. The IR spectrum shows...
-
At one time Mattel proposed acquiring Fisher-Price for $1.2 billion. At the time, Mattel was a major player in the toy industry with 11 percent of the market. Fisher-Price had 4 percent. The other...
-
Explain each of the following phenomena using the invisible hand or social or political forces: a. Firms often pay higher than market wages. b. Wages dont fluctuate much as unemployment rises. c. Pay...
-
Eight cents of every dollar spent at retail stores in America is spent at Walmart. With such market power, Walmart is able to name the price at which it is willing to buy goods from suppliers. a....
-
A teen subminimum training wage law allows employers to pay teenagers less than the minimum wage. a. What effect would you predict this law has, based on standard economic theory? b. In analyzing the...
-
Radical economists argue that labor markets are governed by nonmarket forces such as discrimination as well as by the supply and demand for labor. As they see it, poverty and inequality are not...
-
Hoehn Manufacturing Company experienced the following accounting events during its first year of operation. With the exception of the adjusting entries for depreciation, assume that all transactions...
-
Clark, PA, has been engaged to perform the audit of Kent Ltd.s financial statements for the current year. Clark is about to commence auditing Kents employee pension expense. Her preliminary enquiries...
-
Discuss the physical origins of the gross selection rules for rotational and vibrational Raman spectroscopy.
-
Suppose that you wish to characterize the normal modes of benzene in the gas phase. Why is it important to obtain both infrared absorption and Raman spectra of your sample?
-
Calculate the ratio of the Einstein coefficients of spontaneous and stimulated emission, A and B, for transitions with the following characteristics: (a) 500 MHz radiofrequency radiation, (b) 3.0 cm...
-
Prepare the income statement like the Proforma Net Operating Income statement based on the actual results of the company.
-
What are the main ethical issues that researchers may encounter when conducting business research and how can they address these issues ?
-
How has womens participation in the Olympics changed over the years? Explain

Study smarter with the SolutionInn App


