The molar enthalpy of a ternary mixture of species a, b, and c can be described by
Question:
The molar enthalpy of a ternary mixture of species a, b, and c can be described by the following expression: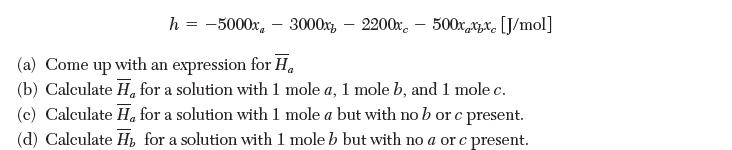
Transcribed Image Text:
h = -5000x - 3000x2200x500xxx [J/mol] (a) Come up with an expression for H (b) Calculate H for a solution with 1 mole a, 1 mole b, and 1 mole c. (c) Calculate H, for a solution with 1 mole a but with no b or c present. (d) Calculate H for a solution with 1 mole b but with no a orc present.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (2 reviews)
Lets tackle each part of the question step by step a To come up with an expression for Ha the partia...View the full answer

Answered By

Pranav Makode
I am a bachelor students studying at professor ram meghe institute of technology and research. I have a great experience of being an expert. I have worked as an expert at helloexperts and solvelancer as a part time job. I have also worked as a doubt solver at ICAD SCHOOL OF LEARNING, which is in Amravati city. I have also worked as an Freelancer.
I have great experience of helping students, as described above. I can help any students in a most simple and understandable way. I will not give you have any chance for complaint. You will be greatfull to accept me as an expert.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Engineering questions
-
1. The molar enthalpy of a ternary mixture of species a, b, and c can be described by the following expression: a. hm = -5000xa - 3000xb - 2200xc-500xaxbxc [J/mol] b. Come up with an expression for...
-
The molar enthalpy of a binary liquid mixture of species 1 and 2 is given by: where T is the temperature in [K]. This expression is valid in the temperature range from 280 K to 360 K. Please answer...
-
Examine the difficulty of adding a proposed lwi.drd, rsl, rs2 (Load With Increment) instruction to MIPS. Interpretation: Reg[rd] = Mem[Reg[rs1] + Reg[rs2]] 1. Which new functional blocks (if any) do...
-
Write a paper that responds to the following: Compare and contrast the legal concepts of "reasonable suspicion" and "probable cause." How does each of these concepts apply to law enforcement...
-
Please complete the 2018 federal income tax return for Tim and Linda Jones. Ignore the requirement to attach the form(s) W-2 to the front page of the Form 1040. If required information is missing,...
-
Assuming that the random process \(U(t)\) is wide-sense stationary, with mean \(\bar{u}\) and variance \(\sigma^{2}\), which of the following functions represent possible structure functions for...
-
Linda Filasky held policies issued by Preferred Risk Mutual Insurance Co. Following an injury in an automobile accident and storm damage to the roof of her home, Filasky sustained loss of income,...
-
What are some of the biggest challenges and changes in trends for Human Resources due to the COVID-19 pandemic which has organizations in flux since 2020 especially with employees working remotely?
-
Plot the partial molar volumes of CO2 and C3H8 in a binary mixture at 100C and 20 bar as a function of mole fraction CO2 using the van der Waals equation of state.
-
Consider a mixture of species 1, 2, and 3. The following equation of state is available for the vapor phase: where, and y1, y2, and y3 are the mole fractions of species 1, 2 and 3, respectively....
-
Simplify each expression. 6 4 5 243 -
-
How do organizational culture and leadership style impact the formulation and execution of strategy, and how can leaders cultivate a strategic mindset and foster alignment throughout the organization...
-
what ways can organizations leverage strategic alliances, mergers, and acquisitions as part of their growth and diversification strategies, while mitigating risks and ensuring synergistic value...
-
Write a function ref_triangular which uses reflections to turn an m X n matrix M into an upper triangular matrix. The output should be a list [Q,R] where is a product of reflections and R is upper...
-
How do organizations effectively balance emergent strategies with deliberate strategies in dynamic environments characterized by uncertainty and rapid change ?
-
Refer to the graph and answer the following questions: Output Time A a. Line A is the typical trend in [(Click to select) b. Line B is the typical trend in (Click to select) B
-
Ethical Issue: Assume that you have drafted a letter to a customer in which you apologize for the way the customers account was fouled up by the Accounting Department. You show the letter to your...
-
we have to compute the letter grades for a course. The data is a collection of student records stored in a file. Each record consists of a name(up to 20 characters), ID (8 characters), the scores of...
-
Describe the mechanism of fluorescence. To what extent is a fluorescence spectrum not the exact mirror image of the corresponding absorption spectrum
-
Describe the principles of laser action, with actual examples.
-
The molar absorption coefficient of a substance dissolved in hexane is known to be 327 dm3 mol-1 cm-1 at 300 nm calculate the percentage reduction in intensity when light of that wavelength passes...
-
public class Q1Test { public static void main(String[] args) { Integer[][] matrix0 = null; Integer[][] matrix1 = {{1}}; Integer[][] matrix2 = {{1, 2, 3}}; Integer[][] matrix3 =...
-
Iterating Over a Collection Iterating a method over a collection of objects is a common task that you may complete unknowingly each day. Within a collection of similar things, you identify the...
-
Problem 8 (10 points). For each of the following methods, determine its big-O characterization in terms of n. A. public static int exampleA (int [ ] arr) { 2 int narr.length, total = 0; Ca public...

Study smarter with the SolutionInn App


