The partial molar volumes of a binary mixture of ethanol (1) and ethylene glycol (2) at 25C
Question:
The partial molar volumes of a binary mixture of ethanol (1) and ethylene glycol (2) at 25°C are reported in the following table. Answer the following questions: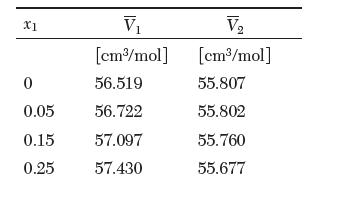
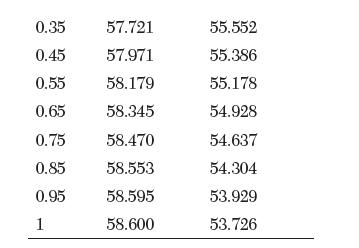

Transcribed Image Text:
V [cm3/mol] 0 56.519 0.05 56.722 0.15 57.097 0.25 57.430 X1 V [cm/mol] 55.807 55.802 55.760 55.677
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (4 reviews)

Answered By

Munibah Munir
I've done MS specialization in finance’s have command on accounting and financial management. Forecasting and Financial Statement Analysis is basic field of my specialization. On many firms I have done real base projects in financial management field special forecasting. I have served more than 500 Clients for more than 800 business projects, and I have got a very high repute in providing highly professional and quality services.I have capability of performing extra-ordinarily well in limited time and at reasonable fee. My clients are guaranteed full satisfaction and I make things easy for them. I am capable of handling complex issues in the mentioned areas and never let my clients down.
4.60+
467+ Reviews
648+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Engineering questions
-
As an undergraduate chemical engineering student, you are involved in a summer research project that requires you to measure the molar volume of a methanol (1) + water (2) binary system at 298.15 K...
-
The molar volume, in [cm3/mol], of a binary mixture of ethanol (1) and ethylene glycol (2) at 25C is given in the following table. Using the graphical method, determine the partial molar volume of...
-
Using the information in Problems 7.13 and 8.20, estimate the heat of vaporization for the first bit of ethanol from ethanol-water solutions containing 25, 50, and 75 mol % ethanol and from a...
-
Based on Exhibit 1, the expected future value of Bond I at maturity is closest to: A. 98.80. B. 103.74. C. 105.00. Lena Liecken is a senior bond analyst at Taurus Investment Management. Kristel...
-
Compare the drug abuse climate in the United States before and after the passage of Prohibition. Video link https://www.youtube.com/watch?v=rIWwHjm-F_o
-
(a) Find the intervals on which f is increasing or decreasing. (b) Find the local maximum and minimum values of f. (c) Find the intervals of concavity and the inflection points. f (x) = sin x + cos...
-
(Lloyd's mirror) A point source of narrowband light is placed at distance \(s\) above a perfectly reflecting planar mirror. At distance \(d\) away, the interference fringes are observed on a screen,...
-
Suppose there are three people in society who vote on whether the government should undertake specific projects. Let the net benefits of a particular project be $150, $140, and $50 for persons A, B,...
-
Discuss how the business make decisions in the context of the behaviour of the various cost of production? Required ( have to be included) : Communicating of cost behaviour from accountant to...
-
Enthalpies of solution, are reported in Table 6.1 for 1 mole of HCl diluted in n moles of H2O at 25C: (a) Consider a mixture of 8 moles H2O and 2 moles HCl. As best you can from these data, estimate...
-
A binary mixture of species a and b behaves as an ideal gas at 300 K and 1 bar. Calculate the partial molar Gibbs energy of species a, Ga, and the total solution Gibbs energy, g, at the following...
-
Trim Corporation acquired 100 percent of Round Corporations voting common stock on January 1, 20X2, for $400,000. At that date, the book values and fair values of Rounds assets and liabilities were...
-
What strategies can be employed to judiciously allocate temporal assets amidst competing priorities, fostering a harmonious balance between personal and professional spheres ?
-
Suppose a bond's price is expected to decrease by 3% if its market discount rate increases by 50 bps. If the bond's market discount rate decreases by 50 bps, the bond price is most likely to change...
-
How does the implementation of intricate time management methodologies facilitate the cultivation of a proactive mindset, thereby catalyzing heightened efficiency and goal attainment ?
-
How do leaders in various fields implement strategies for delegating responsibilities and empowering team members to enhance collective time management capabilities within organizations ?
-
Trembeld Ltd. was developing a new product, and the following timeline occurred during 2015: Required: a. How would Trembeld account for the costs above if the company followed ASPE? b. How would...
-
How can a writer motivate action in a sales letter?
-
What is the role of business risk analysis in the audit planning process?
-
The algebraic forms of the [orbitals are a radial function multiplied by one of the factors (a) z (5z2 - 3r2), (b) y (5y2 - 3r2), (c) x (5x2 - 3r2), (d) Z(x2 Y2), (e) Y(x2- z2), (f) x (z2 y2), (g)...
-
The NO2, molecule belongs to the group C2v' with the C2 axis bisecting the ONO angle. Taking as a basis the N2s, N2p, and 02p orbitals, identify the irreducible representations they span, and...
-
The phenanthrene molecule (29) belongs to the group C2v with the C, axis in the plane of the molecule. (a) Classify the irreducible representations spanned by the carbon 2pz orbitals and find their...
-
What legal or ethical matters related to the legislation is your organization concerned about, if any? Explain your answer and provide examples, if appropriate.
-
A mortgage for a condominium had a principal balance of $ 4 2 , 5 0 0 that had to be amortized over the remaining period of 5 years. The interest rate was fixed at 5 . 2 2 % compounded semi -...
-
How would the risk of customers failing to pay for their purchases on credit influence audit strategy and procedures?

Study smarter with the SolutionInn App


