The vapor pressure of silver (between 1234 K and 2485 K) is given by the following expression:
Question:
The vapor pressure of silver (between 1234 K and 2485 K) is given by the following expression: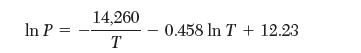
with P in torr and T in K. Estimate the enthalpy of vaporization at 1500 K. State the assumptions that you make.
Transcribed Image Text:
In P= 14,260 T 0.458 In T + 12.23
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Engineering questions
-
do the following,..... Write program that reads a person's first and last names, separated by a space. Then the program outputs last name, comma, first name. Create program that takes in user input...
-
A new and relatively unstudied compound is being investigated for its potential use as a refrigerant in a vapor compression cycle that operates with T = 25C in the boiler and T = 35C in the...
-
The vapor pressure of water at temperature T (in kelvins) is the atmospheric pressure P at which no net evaporation takes place. Use the following table to estimate P²(T ) for T = 303, 313, 323,...
-
Prepare journal entries to record each of the following transactions. The company records purchases using the gross method and a perpetual inventory system. June 1 9 Purchased merchandise with a...
-
The Vanderbilt University Medical Center is representative of many health care delivery systems in the United States today. Even with its long history of success, Vanderbilt faced a $250 million...
-
Jamal and Stephanie each have school loans issued from the same two banks. The amounts borrowed and the monthly interest rates are given next (interest is compounded monthly): (a) Write a matrix A...
-
What are the three items that should be verified before condemning a rotor in the field as having bad rotor bars?
-
The Rustic Welt Company is proposing to replace its old welt-making machinery with more modern equipment. The new equipment costs $10 million and the company expects to sell its old equipment for $1...
-
Explain what is an organizational code of ethics (Ch 3 topic)? Why should organizations have a code of ethics? Explain the "three nudge systems" discussed in the media clip. Explain why or why not,...
-
At a temperature of 60.6C, benzene exerts a saturation pressure of 400 torr. At 80.1C, its saturation pressure is 760 torr. Using only these data, estimate the enthalpy of vaporization of benzene....
-
You wish to know the melting temperature of aluminum at 100 bar. You fi nd that at atmospheric pressure, Al melts at 933.45 K and the enthalpy of fusion is: Heat capacity data are given by: Take the...
-
For the beam problem in Example 8.3, the boundary conditions required that the resultant normal force vanish at each end (x = l). Show, however, that the normal stress on each end is not zero, and...
-
The directors of Sharma Limited are considering three alternative investment proposals, details of which are as follows. Project RENU Project RICHA Project RADHA Initial investment 200,000 240,000...
-
The assessment task is about the egg production industry in Singapore. Currently, there are three egg producers in Singapore, i.e., Seng Choon Farm , Chew's Agriculture , and N&N Agriculture . Your...
-
What is the relationship between current profitability and future profitability and profit growth? Discuss.
-
You estimate that inflation in Mexico will be 6.8% and inflation in the U.S. will be 2.2% from end-2016 to end-2017. Using these and the actual Mexican-U.S. real exchange rate in end-2016, how much...
-
A municipality has issued a long-term municipal bond which indicates that the state legislature will provide support if the bond goes into default. What type of municipal bond offers this protection?
-
Writing Improvement Exercises: Message Openers Your Task. Revise the following e-mail and memo openers so that they are more direct. a. A memo announcing a new procedure: It has come to our attention...
-
Imagine you are the HR manager at a company, and a female employee came to you upset because she felt a male coworker was creating a hostile work environment by repeatedly asking her out on dates...
-
What is the temperature of a two-level system of energy separation equivalent to 300 cm-I when the population of the upper state is one-half that of the lower state?
-
Calculate the ratio of the translational partition functions of xenon and helium at the same temperature and volume.
-
A certain atom has a doubly degenerate ground level, a triply degenerate electronically excited level at 1250 cm3, and a doubly degenerate level at 1300 cm3. Calculate the partition function of these...
-
3. Rank the sites based on the data provided here through Equivalent Property Damage Only (EPDO) average crash frequency and Relative Severity Index (RSI) methods and compare the results. All the...
-
1.1 Meaning of the fields Please read the textbook's section "Revised Sequence ADT-Design Suggestion" for information on the fields. There is a slight difference with the "precursor" pointer. The...
-
Modify the CellPhone class so that it throws the appropriate exception when any of these errors occurs. public class CellPhone { private String model; private String manufacturer; //...

Study smarter with the SolutionInn App


