Use the RedlichKwong equation to calculate the size of vessel you would need to contain 30 kg
Question:
Use the Redlich–Kwong equation to calculate the size of vessel you would need to contain 30 kg of acetylene mixed with 50 kg of n-butane at 30 bar and 450 K. The binary interaction coeffi cient is given by k12 = 0.092.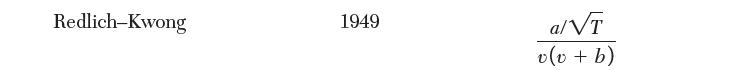
Transcribed Image Text:
Redlich-Kwong 1949 a/VT v(v + b)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)

Answered By

Qurat Ul Ain
Successful writing is about matching great style with top content. As an experienced freelance writer specialising in article writing and ghostwriting, I can provide you with that perfect combination, adapted to suit your needs.
I have written articles on subjects including history, management, and finance. Much of my work is ghost-writing, so I am used to adapting to someone else's preferred style and tone. I have post-graduate qualifications in history, teaching, and social science, as well as a management diploma, and so am well equipped to research and write in these areas.
4.80+
265+ Reviews
421+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Engineering questions
-
A mixture of 60% ethylene and 40% acetylene by moles is at 6 MPa, 300 K. The mixture flows through a preheater where it is heated to 400 K at constant P. Using the Redlich Kwong equation of state...
-
Calculate Z. HR. and SR by the Redlich / Kwong equation for one of the following, and compare results with values found from suitable generalized correlations: (a) Acetylene at 300 K and 40 bar. (b)...
-
Portray in words what transforms you would have to make to your execution to some degree (a) to accomplish this and remark on the benefits and detriments of this thought.You are approached to compose...
-
Generate a matrix of random integer temperatures in Fahrenheit from 70 to 100 for 10 weeks (rows) and 7 days per week (columns). The result should look something like this. Assume the first column is...
-
Which of the following errors would be detected at the point of listing a trial balance? (a) The bookkeeper enters a cash sale as a debit of 49 in the cash book and as a credit of 94 in the sales...
-
The Chair Company provides a 120-day parts-and-labor warranty on all merchandise it sells. The Chair Company estimates the warranty expense for the current period to be $2,650. During this period, a...
-
Figure P7.79 shows the \(60{ }^{\circ} \mathrm{F}\) water flow rates from the branches of a main supply line. Find the total pressure drop \(\left(p_{A}-p_{E} ight)\) for soldered copper pipe. Assume...
-
Little Books Inc. recently reported $3 million of net income. Its EBIT was $6 million, and its tax rate was 40%. What was its interest expense? (Hint: Write out the headings for an income statement...
-
In November, Trinity Donuts incurred $8850 in total costs related to the production of 3500 donuts. In December, Trinity incurred $8600 in total costs. Total fixed costs are $1850 per month. By how...
-
You wish to use the RedlichKwong equation of state to describe a mixture of carbon dioxide (1) and toluene (2). To be as accurate as possible with the mixing rules, you want to include the binary...
-
Using the generalized compressibility charts, calculate the molar volume of ammonia at 92C and 306.5 bar. What phase is ammonia in? GENERALIZED COMPRESSIBILITY CHARTS The principle of corresponding...
-
Find the value of X(0.3) for the initial-value problem using Eulers method with steps of h = 0.1. d.x dt = x - 2t, x(0) = 1
-
1. Assume the following information on a mutual fund: assets: $3 million; liabilities: $500,000; number of shares outstanding: 1,500,000. Compute the funds NAV. 2. A fund started the year with a NAV...
-
Consider a 4% annual coupon bond with a face value of $1000, a yield to maturity of 6% and five years to maturity. Using the formula below calculate the price of the five-year bond: F P (1- (1+0)) +...
-
Megan's company wants to use Google accounts to allow users to quickly adopt their web application. What common cloud federation technologies will Megan need to implement?
-
what ways do visionary leaders leverage emotional intelligence and interpersonal skills to inspire and motivate their teams, fostering a sense of purpose and commitment that transcends conventional...
-
Continuing the previous problem, what is the present value of this perpetuity if the first payment of $100 per year will not be received until five years from now, assuming that subsequent $100...
-
Imagine that you have been selected to compete with students from other universities in presenting a case considering whether the FASB should be abolished and its standard-setting role taken over by...
-
The time to assemble the first unit on a production line is 10 hours. The learning rate is 0.94. Approximately how long will it take for the seventh unit to be assembled? The number of hours needed...
-
How many signals would you expect to see in the 13C NMR spectrum of each of the following compounds? (a) Propylbenzene (d) 1, 2, 4-Trimethylbenzene (b) Isopropylbenzene (e) 1, 3, 5-Trimethylbenzene...
-
Consider carbons x, y, and z in p-methylanisole. One has a chemical shift of 20 ppm; another has 55 ppm, and the third 157 ppm. Match the chemical shifts with the appropriate carbons.
-
To which of the compounds of Problem 13.12 does the 13C NMR spectrum of Figure 13.22 belong?
-
ESSAY: Practicing self-compassion towards the self can be difficult since we often have high standards for ourselves. In this exercise, write an essay detailing your promise to take care of yourself...
-
What are the three possible ways in which gender socialization affected your sexual scripts?
-
for 20 years. Then you are able to increase the contribution to $240 per month for another 20 years. Given a 8.0 percent interest rate, what is the value of your retirement plan after 40 years?

Study smarter with the SolutionInn App


