Use the van der Waals equation of state to plot the inversion line for N2 on a
Question:
Use the van der Waals equation of state to plot the inversion line for N2 on a PT diagram, as schematically shown in Figure 5.10.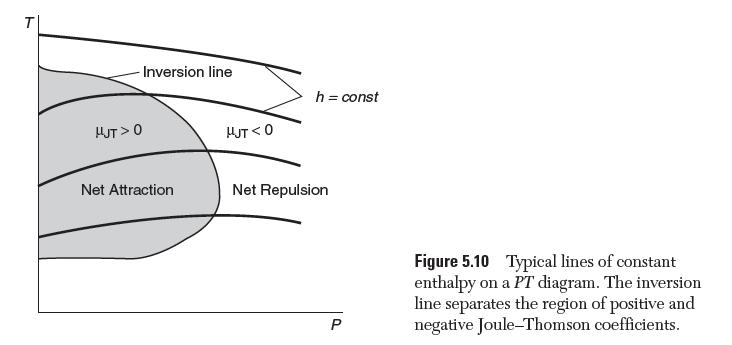
Transcribed Image Text:
T -Inversion line HJT> 0 Net Attraction HUT <0 h = const Net Repulsion P Figure 5.10 Typical lines of constant enthalpy on a PT diagram. The inversion line separates the region of positive and negative Joule-Thomson coefficients.
Step by Step Answer:

This question has not been answered yet.
You can Ask your question!
Related Book For 

Question Posted:
Students also viewed these Engineering questions
-
The overall goal of this problem is to compute the PV and PT equilibrium diagramsfor a single component fluid described by the van derWaals equation of state. Let us recall the key things we need to...
-
The Joule-Thomson coefficient, , given by is a function of temperature. The temperature at which = 0 is known as the inversion temperature. a. Use the van der Waals equation of state to determine...
-
Now assume that there are 1,500 identical firms in this competitive industry. That is, there are 1,500 firms, each of which has the cost data shown in the table. Complete the industry supply schedule...
-
Simplify each expression in Problems 722. Classify each answer by number of terms and degree. (x + 2y3z) - (x - 5y + 4z)
-
For this assignment you are going to construct a basic break-even analysis spreadsheet following the directions from this blog on techrepublic.com:...
-
Solve the Yule-Walker equations provided by Equation (7.58) for the ARMA system given in Example 7.3, assuming a unit-variance white-noise input. Example 7.3. Approximate the ARMA system by an...
-
Review the list of common closing mistakes in the chapter. Give specific examples of how each might affect your success in a sales call.
-
Doe Creek Interior Decorators issued a 120-day, 7% note for $150,000, dated February 18, 2014, to La Fleur Furniture Company on account. a. Determine the due date of the note. b. Determine the...
-
A project has an initial cost of $35,000, expected net cash inflows of $14,000 per year for 11 years, and a cost of capital of 10%. What is the project's PI? (Hint: Begin by constructing a time...
-
Ethylene is liquefi ed by a JouleThomson expansion. It enters the throttling process at 50 bar and 0C and leaves at 10 bar. What is the fraction of the inlet stream that is liquefi ed?
-
Determine JT for steam at 1 MPa and 300C using data from the steam tables.
-
Consider the unity feedback system shown in Figure P9.1 with a. Find the location of the dominant poles to yield a 1.2 second settling time and an overshoot of 15%. b. If a compensator with a zero at...
-
An airline company is considering giving rates reduced by up to 50 per cent to members of the families of businesspeople at certain times of the week, in order to encourage businesspeople to take...
-
Jennifer did not file a tax return for 2006 because she honestly believed that no tax was due. In 2017, the IRS audits Jennifer and the agent proposes a deficiency of $500.
-
Harry and Silvia, a married couple, are both age 67 and legally blind. What is their standard deduction for 2017?
-
Explain why a random sample is one of the best ways to obtain a sample that is representative of the population.
-
Michaels adjusted gross income for 2017 is $90,000. He is age 30 and single with no dependents. What is Michaels taxable income?
-
Explain the difference between a CAFR and general purpose external financial reports.
-
Suppose the S&P 500 futures price is 1000, = 30%, r = 5%, = 5%, T = 1, and n = 3. a. What are the prices of European calls and puts for K = $1000? Why do you find the prices to be equal? b. What...
-
Use eqn 19.27 to deduce expressions for (a) The root mean square separation of the ends of the chain, (b) The mean separation of the ends, and (c) Their most probable separation. Evaluate these three...
-
Evaluate the radius of gyration, Rg, of (a) A solid sphere of radius a, (b) A long straight rod of radius a and length I. Show that in the case of a solid sphere of specific volume v" Rg/nm =...
-
Calculate the excluded volume in terms of the molecular volume on the basis that the molecules are spheres of radius a. Evaluate the osmotic virial coefficient in the case of bushy stunt virus, a =...
-
Lana is filling out an application form for the position of an elementary school teacher While she is thing it out, she comes across section that asks for information about her high school and...
-
What time does the medicom make to their source and strategy for melt - blown during the COVID - 1 9 pandemic and why was Canada at particular challenge from Medicom.
-
How would you improve the master baker's productivity? How will you improve the bread factory's productivity?

Study smarter with the SolutionInn App


