Using data from Table 4.2, estimate the equilibrium bond length that would exist in a molecule of
Question:
Using data from Table 4.2, estimate the equilibrium bond length that would exist in a molecule of Xe2.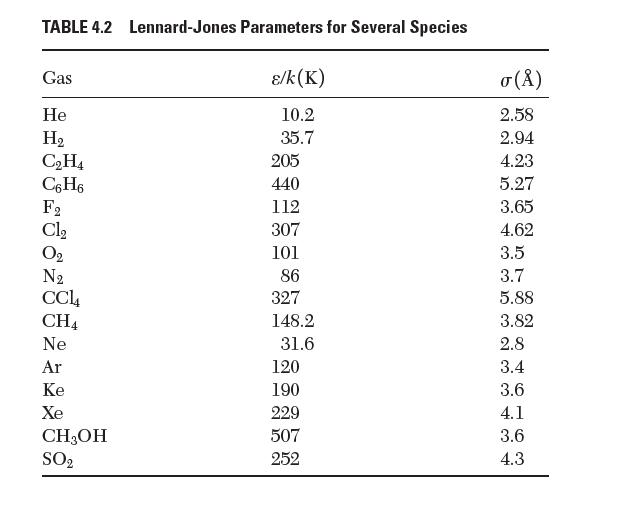
Transcribed Image Text:
TABLE 4.2 Lennard-Jones Parameters for Several Species /k(K) 10.2 35.7 Gas He H CH4 C6H6 288 28 Cl CH4 Ne Ar Ke Xe CH3OH SO 205 440 112 307 101 86 327 148.2 31.6 120 190 229 507 252 () 2.58 2.94 4.23 5.27 3.65 4.62 3.5 3.7 5.88 3.82 2.8 3.4 3.6 4.1 3.6 4.3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)

Answered By

JAPHETH KOGEI
Hi there. I'm here to assist you to score the highest marks on your assignments and homework. My areas of specialisation are:
Auditing, Financial Accounting, Macroeconomics, Monetary-economics, Business-administration, Advanced-accounting, Corporate Finance, Professional-accounting-ethics, Corporate governance, Financial-risk-analysis, Financial-budgeting, Corporate-social-responsibility, Statistics, Business management, logic, Critical thinking,
So, I look forward to helping you solve your academic problem.
I enjoy teaching and tutoring university and high school students. During my free time, I also read books on motivation, leadership, comedy, emotional intelligence, critical thinking, nature, human nature, innovation, persuasion, performance, negotiations, goals, power, time management, wealth, debates, sales, and finance. Additionally, I am a panellist on an FM radio program on Sunday mornings where we discuss current affairs.
I travel three times a year either to the USA, Europe and around Africa.
As a university student in the USA, I enjoyed interacting with people from different cultures and ethnic groups. Together with friends, we travelled widely in the USA and in Europe (UK, France, Denmark, Germany, Turkey, etc).
So, I look forward to tutoring you. I believe that it will be exciting to meet them.
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Engineering questions
-
Northern Virginia Community College HOW MUCH FINANCIAL RISK SHOULD YOU TAKE? Mark D. D'Antonio Nova Southeastern University FORT LAUDERDALE, FLORIDA, U.S.A. Abstract A successful retirement...
-
Using data from Table 8.4 on bond enthalpies, show that the more C---H bonds a molecule has compared to C-----O and O----H bonds, the more energy it can store.
-
PLEASE GIVE CORRECT ANSWERS Prove that the number of comparators in any sorting network is (n log n). [4 marks] (ii) What does Part (d)(i) imply in terms of the depth of any sorting network? [1 mark]...
-
1. By integrating Planck's codiation law over all wave hengths power radiated per square meter of a is given by: R(T) = (55) T" proof that the cavity's surface note: x= he AKT 2 xdx 3 % ex-1
-
John Timms is the sole owner of Sunshine Wholesale Traders, a company which buys fruit from farmers and sells it to supermarkets. All goods are collected from farms and delivered to supermarkets on...
-
What are the factors the courts look at to determine whether there is a duty?
-
The Wide World of Fluids article titled "New Hi-tech Fountains,". The fountain shown in Fig. P8.55 is designed to provide a stream of water that rises \(h=10 \mathrm{ft}\) to \(h=20 \mathrm{ft}\)...
-
a. Evaluate each of these alternatives. On the basis of the limited information presented, recommend the one you feel is best. b. If Casinos International's stock price rises to $60, what will happen...
-
Jim Carrie Company shows a balance of $221,140 in the Accounts Receivable account on December 31, 2025. The balance consists of the following. Installment accounts due in 2026 $23,000 Installment...
-
Calculate the van der Waals parameter b for CH4, C6H6, and CH3OH. Based on these values, estimate the molecular diameter of each species. Compare the values obtained with those in Table 4.2. TABLE...
-
Calculate the bond strength in [eV] of a sodium ion in a crystal of NaCl. For the salt lattice: (a) Consider only the six nearest-neighbor Cl- ions. The Cl- ions are at a distance r = 2.76 . from the...
-
What factors affect consumer selection of a payment instrument?
-
Analyze the changes in Nowcorp's financial position from the beginning to the end of the year Unknown node type: p " id="MathJax-Element-5-Frame" style="font-size: 121%; position: relative;"...
-
My clients Cosmo and Wanda are considering buying a Mansion, and they have successfully negotiated the price down to $275,000. They plan on making a down payment of 20% and financing the remainder...
-
Describe the difference between growth that occurs as a result of an increase in inputs and growth that occurs as a result of an increase in output per input.
-
Provide a good example of a company that has a large positive net cash from operating activities? B. Provide a good example of a company that has negative net cash from operating activities?
-
The investment advisor recommended looking into managed funds, which could help remove from Jamie Lee and Ross the burden of decision making about when to buy and sell. But Ross was considering index...
-
For each of the following independent situations, identify the relevant objective(s) of financial reporting that the company could be overlooking. Discuss each of these objectives. 1. The president...
-
What are the main distinctions between the different schools of legal interpretation?
-
Is either of the epoxides formed in the preceding reactions chiral? Is either epoxide optically active when prepared from the alkene by this method?
-
What is the principal organic product formed in the reaction of ethylene oxide with each of the following? (a) Sodium cyanide (NaCN) in aqueous ethanol (b) Sodium azide (NaN3) in aqueous ethanol (c)...
-
Given the starting material 1-methyl-1, 2-epoxycyclopentane, of absolute configuration as shown, decide which one of the compounds A through C correctly represents the product of its reaction with...
-
Mr. X wants to borrow some books from the library. He is carrying a bag with capacity of W(kg). Each of the books in the library has a fixed weight. Multiple copies of the same books have the same...
-
Cara's team does not have enough funding for their assigned project. She approaches leadership to explain their situation and request an extension of their budget. In this scenario, Cara is...
-
d. If a portfolio of the two assets has a beta of 1.24, what are the portfolio weights? (A negative value should be indicated by a minus sign. Do not round intermediate calculations. Enter your...

Study smarter with the SolutionInn App


