Verify that the standard half-cell potentials reported in Table 9.1 for the reactions between cupric, cuprous, and
Question:
Verify that the standard half-cell potentials reported in Table 9.1 for the reactions between cupric, cuprous, and solid copper![]()
and![]()
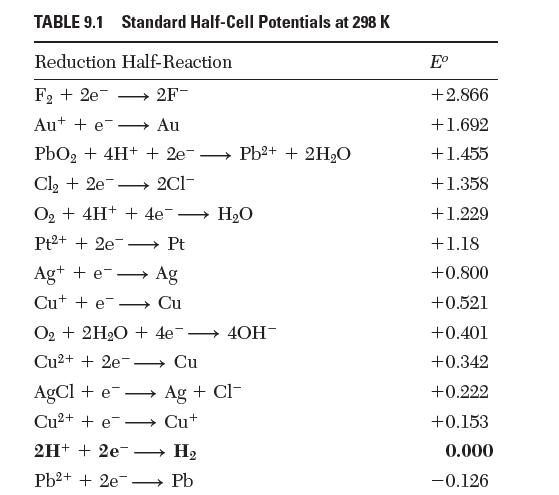
Transcribed Image Text:
Cu+e Cut, Cu+ + 2 e Cu,
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (1 review)

Answered By

John Kimutai
I seek to use my competencies gained through on the job experience and skills learned in training to carry out tasks to the satisfaction of users. I have a keen interest in always delivering excellent work
4.70+
11+ Reviews
24+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Engineering questions
-
Verify that the standard matrix of the projection onto W in Example 7.31 (as constructed by Theorem 7.11) does not depend on the choice of basis. Take as a basis for W and repeat the calculations to...
-
Verify that the standard matrix of the projection onto W in Example 7.31 (as constructed by Theorem 7.11) does not depend on the choice of basis. Take as a basis for W and repeat the calculations to...
-
What is the effect of a viscosity (competence) difference between strain markers and the matrix?
-
How much typical storage redundancy do cloud storage providers like Dropbox, iCloud Drive, Google Drive and OneDrive use? In other words, how do cloud storage vendors prevent files from getting lost...
-
The pressure, volume, and temperature of a mole of an ideal gas are related by the equation PV = 8.31T, where P is measured in kilopascals, V in liters, and T in kelvins. Use differentials to find...
-
Organize the data set in Example 1 using a stem-and-leaf plot that has two rows for each stem. Describe any patterns. Data from Example 1 The data set at the left lists the numbers of text messages...
-
Harlen Industries has a simple forecasting model: Take the actual demand for the same month last year and divide that by the number of fractional weeks in that month. This gives the average weekly...
-
a. Since year 2000 STI share price had been trading below $5/share with 10-year options. Based on the Option Pricing Model and with an estimated growth of 5%, the future estimated stock price would...
-
Hydrogen-based fuel cells show promise as an alternative fuel source. They use a galvanic cell in which oxygen gas is supplied to one compartment and hydrogen gas to another. The reduction of O2 gas...
-
Electrolysis of NaCl is used to manufacture NaOH, Cl2, and H2. Answer the following questions: (a) Determine the overall reaction and each half-cell reaction. (b) Write the process in terms of...
-
Treating the data as samples from larger populations, test the claim that there is a difference between the mean for presidents and the mean for British monarchs. Presidents 10 29 26 28 15 23 17 25 0...
-
A company can purchase a machine, which would yield net revenues of 5 0 , 0 0 0 pa . over the first two years and then 4 0 , 0 0 0 p . a . for a further two years. After these four years of...
-
Midland Energy Resources, Inc" Assignment Questions: How are Mortensen's estimates of Midland's cost of capital used? How, if at all, should these anticipated uses affect the calculations? Calculate...
-
What strategy has been found to be particularly helpful for keeping teams together?
-
Determine the domain and range of the functions below. f(x, y) = [x/y4] for any real numbers x and y where y + 0. b. The function that assigns to a bit string the numerical position of the last 0 in...
-
What is r? Remember, r is just the distance from the point at which we are calculating the electric field (which is point P) to the place where the differential charge dq is located (which is at x)....
-
What are seven goals of ethical business communicators?
-
You are planning to purchase your first home five years from today. The required down payment will be $50,000. You currently have $20,000. but you plan to contribute $500 each quarter to a special...
-
Studies of combustion reactions depend on knowing the concentrations of H atoms and HO radicals. Measurements on a flow system using EPR for the detection of radicals gave information on the...
-
I.D. Chapple-Sokol, Cl Giunta, and R.G. Gordon (J Electrochem Sac 136,2993 (1989)) proposed the following radical chain mechanism for the initial stages of the gas-phase oxidation of silane by...
-
For many years the reaction Hz (g) + I2 (g) →7 2 HI (g) and its reverse were assumed to be elementary bimolecular reactions. However, I.H. Sullivan (J. Chem. Phys. 46, 73 (1967)) suggested that...
-
Solve the Question 2100 2800 3500 Fixed Variable Total Cost Cost per smothie served What is the Total Fixed cost $2,500.00 Variable Cost $0.75
-
A virtual classroom forum is a place for asynchronous online discussion where students and the teacher contribute ideas, options, knowledge and clarify doubts on a particular topic. Approach Tell us...
-
Consider the following evaluation and control tools used by Dana Shelton, a marketing manager at an insurance company, to evaluate the insurer's agencies: Tool AMs. Shelton set a range of acceptable...
Easy Computer Basics Windows 7 See It Done Do It Yourself 1st Edition - ISBN: 0789742276 - Free Book

Study smarter with the SolutionInn App


