What is the composition of vapor that is in equilibrium with a liquid mixture with the following
Question:
What is the composition of vapor that is in equilibrium with a liquid mixture with the following composition at 250 K? What is the pressure? Assume ideal behavior.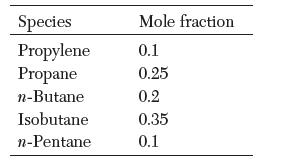
Transcribed Image Text:
Species Propylene Propane n-Butane Isobutane n-Pentane Mole fraction 0.1 0.25 0.2 0.35 0.1
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)

Answered By

Madhur Jain
I have 6 years of rich teaching experience in subjects like Mathematics, Accounting, and Entrance Exams preparation. With my experience, I am able to quickly adapt to the student's level of understanding and make the best use of his time.
I focus on teaching concepts along with the applications and what separates me is the connection I create with my students. I am well qualified for working on complex problems and reaching out to the solutions in minimal time. I was also awarded 'The Best Tutor Award' for 2 consecutive years in my previous job.
Hoping to get to work on some really interesting problems here.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Engineering questions
-
The table below shows the vapor pressures, VP (kPa) for nhexane and noctane. (1) Assuming ideal solution and gas behavior, construct Txy and xy diagrams for this system at 101 kPa. (2) When a liquid...
-
A liquid mixture containing 25 mol% benzene and 75 mol% ethyl alcohol, in which components are miscible in all proportions, is heated at a constant pressure of 1 atm (101.3 kPa, 760 tort) from a...
-
Tell whether the given side lengths of a triangle can represent a right triangle. 36, 48, and 60
-
From the following facts, calculate what Adam Dell must pay Black Co. for the purchase of a bedroom set. Sale terms are 2/10, n/30. a. Invoice price before tax, $5,000, dated April 5. b. Returned one...
-
John Hildreth was the sole shareholder, director, and officer of a New Jersey corporation, HCE Inc., also referred to as HCE-NJ. Engaged in the construction business, HCE-NJ began to do business in...
-
Derive Equations 5.26 and 5.30 . == Ef10f1Vf+ Em1m1Vm_ Ef1ff + Em1m1vm 1 Ef1Vf+ Em1Vm (5.26)
-
Berry Company sold goods with a total selling price of $800,000 during the year. It purchased goods for $380,000 and had beginning inventory of $67,000. A count of its ending inventory determined...
-
Spielman Leather, Inc., produces three sizes of sports gloves: small, medium, and large. A glove pattern is first stenciled onto leather in the Pattern Department. The stenciled patterns are then...
-
A liquid mixture containing 40% cyclohexane, 20% benzene, 25% toluene, and 15% n-heptane is in equilibrium with its vapor at 1 bar. Determine the temperature and the vapor composition.
-
What is the lowest temperature to which a vapor mixture of 1 mole n-pentane and 2 moles n-hexane at 1 bar can be brought without forming liquid? Assume the liquid forms an ideal solution.
-
Using the data from Exercise 29, find the distance traveled by the planet Mercury during one complete orbit around the Sun. (If your calculator or computer algebra system evaluates definite...
-
Finally, turn in the required graphical or numerical results for simulation problems 1) through 4). 1) Generate a probability histogram of the number of rolls required of two dice before a sum of "7"...
-
Forecast Revenue correctly based on this? Multiperiod Forecast Revenue Costs of goods sold Bakery-caf Dough sold to franchisees Depreciation General and administrative Operating profit Interest...
-
Based on a simple random sample of 100 observations from a population, it is found that p = 0.68. Use this sample and answer each of the following questions. (1) Test Ho p = 0.6 against H p > 0.6....
-
1. Please describe clearly the application of the Library Information System diagram in a School library. Complete with examples of each subsystem in the those diagrams! 2. Look for some examples of...
-
1. Describe how the screened - host firewall provides security and protectionIdentify how the firewall could be compromised 2. Recommend security measures to mitigate the potential of compromise 3....
-
Find estimates of price elasticity for a variety of consumer goods and services. Explain what price elasticities of 0.5 and 2.4 mean.
-
If the amplifier indicated by the box input impedance of oo, which of the following statements are true ? has an open loop gain as well as Feedback factor (\beta = 1/ R_1\) The feedback is voltage...
-
On a cold, dry morning after a frost, the temperature was -5C and the partial pressure of water in the atmosphere fell to 0.30 kPa. Will the frost sublime? What partial pressure of water would ensure...
-
Given that Cp = T(V/T)p - V, derive an expression for .u in terms of the van der Waals parameters a and b, and express it in terms of reduced variables. Evaluate u at 25C and 1.0 atm, when the molar...
-
The normal boiling point of hexane is 69.0C. Estimate (a) Its enthalpy of vaporization and (b) Its vapour pressure at 25C and 60C.
-
A husband and his wife are killed in an accident. Their estate is probated and after all the bills are paid, it nets out at $ 1,350,000. At a combined federal and state tax rate of 10%, how much...
-
You need to calculate the completed and removed from WIP Inventory - Mixing. Costs in beginning WIP inventory $1,700 Additional costs to finish units in beginning WIP inventory $400 Costs of units...
-
Debbies beauty boutique had cost for april of $9800, and earned a profit of $3250 in April. What was the total sales and revenue brought in by Debbie in april?

Study smarter with the SolutionInn App


