You are using the PengRobinson equation of state to determine the entropy change of an ideal gas:
Question:
You are using the Peng–Robinson equation of state to determine the entropy change of an ideal gas: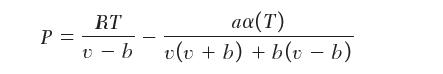
Is it better to try s = s (T,v) or s = s (T,P)? Explain.
Transcribed Image Text:
P = RT v-b aa(T) v(v + b) + b(v - b)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)

Answered By

Akshay Singla
as a qualified engineering expert i am able to offer you my extensive knowledge with real solutions in regards to planning and practices in this field. i am able to assist you from the beginning of your projects, quizzes, exams, reports, etc. i provide detailed and accurate solutions.
i have solved many difficult problems and their results are extremely good and satisfactory.
i am an expert who can provide assistance in task of all topics from basic level to advance research level. i am working as a part time lecturer at university level in renowned institute. i usually design the coursework in my specified topics. i have an experience of more than 5 years in research.
i have been awarded with the state awards in doing research in the fields of science and technology.
recently i have built the prototype of a plane which is carefully made after analyzing all the laws and principles involved in flying and its function.
1. bachelor of technology in mechanical engineering from indian institute of technology (iit)
2. award of excellence in completing course in autocad, engineering drawing, report writing, etc
4.70+
48+ Reviews
56+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Engineering questions
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-6. On December 12, Irene purchased the building where her store is located. She paid...
-
On 01/01/2019 ABC Company purchase a truck for $140,000. The truck has a $50,000 salvage value and a life of 4 years with estimated total miles of 200,000. Fill in the answers below: 1. How much is...
-
A company had average total assets of $500,000, gross sales of $575,000, and net sales of $550,000. The companys total asset turnover is a. 1.15. b. 1.10. c. 0.91. d. 0.87. e. 1.05.
-
Choose three different presidents from Harry S. Truman through Barack Obama and look at the data. www.presidency.ucsb.edu/data/popularity.php?pres=44 For this, navigate to website's collection of job...
-
How many different vertical arrangements are there of 10 flags if 5 are white, 3 are blue, and 2 are red? Approach Because there are nondistinct items and order matters, use the formula for finding...
-
How do you mine use cases to find classes?
-
State whether or not a trust is created in each of the following situations: (a) A declares herself trustee of the bulk of my securities in trust for B. (b) A, the owner of Blackacre, purports to...
-
If the indirect method is used to calculate the intrinsic value of a company's equity. How to compare the intrinsic value per share and the value per share?
-
Consider a gas that undergoes a process from state 1 to state 2. You know the ideal gas heat capacity and an equation of state. Which of the following hypothetical paths would be most appropriate to...
-
Of the following mixture, which do you think has entropy departure function of larger magnitude (a) 50 mol% methane mixed with 50 mol% ethane (b) 50 mol% acetone mixed with 50 mol% chloroform?...
-
A university found that 20% of its students withdraw without completing the introductory statistics course. Assume that 20 students registered for the course. a. Compute the probability that 2 or...
-
Some people believe that the rise of regional trading blocs threatens free trade progress made by the World Trade Organization (WTO). What arguments can you present to counter this point of view? Do...
-
If the age at death of a 20-year-old U.S. woman is normally distributed with a mean of 76.4 years and a standard deviation of 8.1 years, what is the probability that she will live past 80?
-
Explain how you would obtain a simple random sample from this population: varsity athletes at your school.
-
What is it called when trade shifts away from nations not belonging to a trading bloc and toward member nations?
-
In his 1868 work, Carl Wunderlich concluded that temperatures above 100.4 Fahrenheit should be considered feverish. In a 1992 study, Maryland researchers suggested that 99.9 Fahrenheit was a more...
-
How does the statement of cash flows under GASB standards differ from the statement of cash flows under FASB standards?
-
The Taylor's series expansion for cosx about x = 0 is given by: where x is in radians. Write a user-defined function that determines cosx using Taylor's series expansion. For function name and...
-
The rate of the reaction A + 3 B --7 C + 2 D was reported as 1.0 mol dm-1 S-1. State the rates of formation and consumption of the participants.
-
The rate of consumption of B in the reaction A + 3 B 7 C + 2 D is 1.0 mol dm3 S-1. State the reaction rate, and the rates of formation or consumption of A, C, and D.
-
The rate law for the reaction in Exercise 22.1b was found to be v = k[A][BF, What are the units of k? Express the rate law in terms of the rates of formation and consumption of (a) A, (b) C.
-
Shell Corporation is a garment manufacturer and is considering installing a sewing machine for $3,800,000. The machine will depreciate straight-line over three years and will have zero salvage value....
-
Solve these below uestions, E. Prepare an overhead budget for July, August and September and in total for the quarter. . Prepare a selling and administrative expenses budget for July, August and...
-
More generally, what is the optimal choice of leverage (L*)?

Study smarter with the SolutionInn App


