Your colleague reports that the conversion to n-butane from the gas phase hydrogenation reaction of 1-butene increases
Question:
Your colleague reports that the conversion to n-butane from the gas phase hydrogenation reaction of 1-butene increases as temperature increases: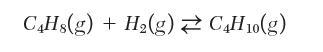
Is this possible? Explain.
Transcribed Image Text:
C4Hs(g) + H(g) C4H0(g)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (2 reviews)
Yes it is possible that the conversion of 1butene to nbutane increases as the temperature increases ...View the full answer

Answered By

Danish Sohail
My objective is to become most reliable expert for clients. For last 10 years I have been associated with the field of accounting and finance. My aim is to strive for best results and pay particular attention to client needs. I am always enthusiastic to help clients for issues and concerns related to business studies. I can work on analysis of the financial statements, calculate different ratios and analysis of ratios. I can critically evaluate stock prices based on the financial analysis and valuation for companies using financial statements of the business entity being valued with use of excel tools. I have expertise to provide effective and reliable help for projects in corporate finance, equity investments, financial accounting, cost accounting, financial planning, business plans, marketing plans, performance measurement, budgeting, economic research, risk assessment, risk management, derivatives, fixed income investments, taxation, auditing, and financial performance analysis.
4.80+
78+ Reviews
112+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Engineering questions
-
a. Example 13-1: Batch Reactor with an Exothermic Reaction Wolfram 1. Adiabatic Case: Use Wolfram to see whether you can find a trajectory that is ready to ignite and whose trajectory looks like a...
-
(a) LEP Table 12-2: Exothermic Reaction with Heat Exchange Download the Polymath, MATLAB, Python, or Wolfram codes for the algorithm and data given in Table T12-2 for the exothermic gas phase...
-
Portray in words what transforms you would have to make to your execution to some degree (a) to accomplish this and remark on the benefits and detriments of this thought.You are approached to compose...
-
A baseball player usually has four at bats each game. Suppose the baseball player is a lifetime 0.25 hitter. Find the probability that this player will have: (a) Two hits out of four at bats (b) No...
-
Why is the price elasticity of demand a relative measure? That is, why is elasticity measured in percentage terms rather than absolute terms? 1. So the coeffecient of elasticity will not be dependent...
-
What about bad debts? Should you make similar adjustments in the cash flow statement for them? Why/why not?
-
Comment on this statement: Dynamic modeling is about interaction.
-
Redfern Corporation, a calendar year taxpayer, has been an S corporation for several years. Rod and Kurt each own 50% of Redferns stock. On July 1 of the current year (assume a non-leap year),...
-
Given the parity matrix H 1 0 1 1 0 1 0 1 1 0 0 H 1 1 00 0 0 01 1 00 00 0 1 0 00 0 1 0 0 0 100 100 000 1 0 1 0 1 0 0 0 0 00000 1- a) Is the given matrix regular or irregular/Explain b) Find the...
-
Which of the following conditions would you use if you needed to develop an industrial process to produce ethanol from acetylene? Explain. (a) 25C and 1 bar. (b) 250C and 1 bar. (c) 25C and 150 bar....
-
Consider the gas phase hydrogenation reaction of propylene to form propane: To increase the equilibrium conversion, would it help to (a) Increase the pressure? (b) Increase the temperature? (c) Add...
-
Compare and contrast the two common-size balance sheets below. Which one do you think may belong to an auto manufacturer? To a computer manufacturer?
-
How does an asset gain or loss develop in pension accounting?
-
Cooper Investments reported an unusual gain from the sale of certain assets in its 2015 income statement. How does intraperiod tax allocation affect the reporting of this unusual gain?
-
At December 31, 2015, Ashley Co. has outstanding purchase commitments for purchase of 150,000 gallons, at 6.20 per gallon, of a raw material to be used in its manufacturing process. The company...
-
Under what circumstances is it appropriate to record goodwill in the accounts? How should goodwill, properly recorded on the books, be written off in order to conform with IFRS?
-
Manchester United, Inc. sold 10,000 season tickets at 2,000 each. By December 31, 2015, 8 of the 20 home games had been played. What amount should be reported as a current liability at December 31,...
-
Identify the budgetary accounts used in federal agency accounting and explain the sequential flow of budgetary authority through the accounts in your own words.
-
1. Use these cost, revenue, and probability estimates along with the decision tree to identify the best decision strategy for Trendy's Pies. 2. Suppose that Trendy is concerned about her probability...
-
Name the functional group(s) present in each of the compounds in Problem 2.17.
-
Name the functional group(s) present in each of the compounds in Problem 2.18.
-
Determine whether these structures represent the same compound orisomers: a) b) @ Y il Q0 CHCH3 So d) OCH,
-
When information is presented in the form of a bar graph or time-series graph, you could get more exact values if all the data were just listed out in table form. Then why not always do that? Why...
-
At what price does a company issue a $4,800,000, 6%, six-year bond if the market rate is 8% and interest is compounded semi-annually?
-
A Government of Canada V39065 issue 90-day T-bill achieved its highest rate of return on May 24, 2000, with a yield of 5.74%. It realized its lowest rate of return on February 26, 2010, with a yield...

Study smarter with the SolutionInn App


