(a) Rank these molecules in order of increasing boiling point: (b) State your reason for the order...
Question:
(a) Rank these molecules in order of increasing boiling point:
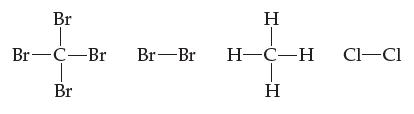
(b) State your reason for the order you chose in part (a).
Transcribed Image Text:
Br Br-C-Br Br Br Br H H-C-H H CI-CI
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
The order of increasing boiling point for the molecules in the image is Bromine Br2 Carbon tetrachlo...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Introductory Chemistry Atoms First
ISBN: 9780321927118
5th Edition
Authors: Steve Russo And Michael Silver
Question Posted:
Students also viewed these Sciences questions
-
(a) Rank these molecules in order of increasing boiling point: (b) State your reason for the order you chose in part (a). H H-C-OH H H-H H H-C-H H H-Cl
-
Rank the following molecules in order of increasing boiling point (without looking up the real values!): (a) 3-methylheptane; (b) Octane; (c) 2,4-dimethylhexane; (d) 2,2,4-trimethylpentane.
-
Rank the following isomers in order of increasing boiling point, and explain the reasons for your order of ranking. OCH2CH2OH CH C 2-(vinyloxy ethanol OCH2CH CH,CH CHC-OH ethyl acetate butyric acid
-
(a) Find the Maclaurin series for the function f(x)= ln(1+x) and hence that for In(1+x) (b) By keeping the first four terms in the Maclaurin series for In(1+x) integrate the function In(1+x) from x =...
-
How does the market-driven approach illustrate that HR has strategic, operational, and administrative roles at SYSCO?
-
If unit 1 requires 200 hours to produce and the labor records for an Air Force contract of 50 units indicates an average labor content of 63.1 hours per unit, what was the learning rate? What total...
-
Use the technique discussed in Section9.4 to develop a GEE approach for zeroinflated Poisson model for count responses in longitudinal studies. Section9.4: 9.4 Marginal Models for Longitudinal Data...
-
Multiple Choice. Choose the best answer. 1. Which of the following correctly states the role of budgeting in proprietary funds? a. Proprietary fund managers should ensure that a valid appropriation...
-
Map the EER model of the following databases, to a relational model. Show all integrity types(Clarify your answer by underline the Primary key, and dotted line the Foreign Key. ( Member SSN {PK}...
-
Show how two CH 2 Cl 2 molecules in the liquid phase are oriented with respect to each other and explain why the molecules align this way.
-
Acetic acid, is very soluble in water because of the many hydrogen bonds that can form. Add water molecules to this drawing and show the hydrogen bonds possible. HC :O:
-
Consider two groups of students: B1, students who received high scores on tests, and B2, students who received low scores on tests. In group B1, 80% study more than 25 hours per week, and in group...
-
B Borko sent a tax invoice to B Andres $1727 ($1570 + $157 GST) four months ago. All attempts to obtain payment have failed and B Andres can no longer be contacted. On 30 June 2022, B Andres is...
-
L Narrabeen mailed a $4400 ($4000 + $400 GST) tax invoice to D Why on 2 April 2022. On 8 April L Narrabeen received a cheque from D Why for the full amount owing. The deposit for that day included D...
-
On 1 January 2021, machinery was purchased for $29 700 ($27 000 + $2700 GST) to be depreciated at 20% p.a. Insurance account was $1200 at 31 March 2021. Annual insurance premium was paid on 1 April...
-
Prepare the accounts receivable and accounts payable control accounts for April for K Ann. Balance as per accounts receivable control account 31 March 2022 Balance as per accounts payable control...
-
Prepare the journals for the dates in August 2022 and post to the general and subsidiary ledgers for S Ireland who operates with perpetual inventory. 2 P Dipstick purchased stock from S Ireland for a...
-
List causes that could lead to a variable overhead efficiency variance?
-
To balance the chemical equation SiH3 + O2 SiO2 + HO, you could introduce coefficients a, b, c, d and write aSiH3 + bO2 cSiO + dHO then write linear equations for each element. The equation for Si...
-
What is a half-life? Is the half-life for a first-order reaction dependent on concentration?
-
In a sequential reaction, what is an intermediate?
-
What is meant by the rate-determining step in a sequential reaction?
-
historical data for precious metal prices, 10- year government bond yields (assumed to be the risk-free rate of interest), and the aggregate equity market index. All data are in monthly frequency....
-
Pepsi is experimenting with a new version of their originalMountain Dew soda flavor and wants to know how it will be receivedby the market. Specifically, they want to know if the new versionwill be...
-
12 3 B PMT472 APPLIED PROJECT MANAGEMENT 2.3 ASSIGNMENT: WEEK 2 EXERCISES A CH. 5 EXERCISE 7 D Omega 2 Project. Using the "complexity weighting" scheme shown in Table 5.2 and the following function...

Study smarter with the SolutionInn App


