Ascorbic acid (otherwise known as vitamin C) prevents scurvy and may even help prevent certain cancers as
Question:
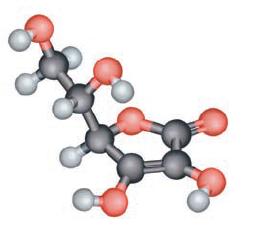
Ascorbic acid (otherwise known as vitamin C) prevents scurvy and may even help prevent certain cancers as well as the common cold. It is composed of 40.92% carbon (C), 4.58% hydrogen (H), and 54.50% oxygen (O) by mass. To the right is a ball-and-stick picture of ascorbic acid. What is the actual molecular formula of vitamin C?
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 33% (3 reviews)
The molecular formula of ascorbic acid vitamin C is C6H8O6 Proof We can use the following steps to c...View the full answer

Answered By

Facundo buzzetti
I have been tutoring for the past 3 years, and during this time I have gained a lot of hands-on experience working with students of various ages and academic levels. I am proficient in tailoring my teaching style to meet the individual needs of each student, whether they learn best through visual aids, hands-on activities, or a combination of different approaches. I also have experience in creating personalized study plans and providing ongoing support to help students achieve their academic goals. Additionally, I have proficiency in using online tools and platforms to facilitate virtual tutoring sessions and adapt to different learning environments.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Introductory Chemistry Atoms First
ISBN: 9780321927118
5th Edition
Authors: Steve Russo And Michael Silver
Question Posted:
Students also viewed these Sciences questions
-
Calculate the percentage by mass of the indicated element in the following compounds: (a) Carbon in acetylene, C2H2, a gas used in welding (b) Hydrogen in ascorbic acid, HC6H7O6, also known as...
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
Due to its severity, bird flu, or avian flu, has caused great concern worldwide. The main drug for its treatment is Tamiflu. Tamiflu is an antiviral drug and acts to minimize the effects of bird flu...
-
The slurry of Prob. 29.3 is to be filtered in a press having a total area of 8 m 2 and operated at a constant pressure drop of 2 atm. The frames are 36 mm thick. Assume that the filter medium...
-
Using the income statement and balance sheet for Kau and Sons Co., compute the three components of return on equity'profitability, efficiency, and leverage'based on the DuPont framework, for the...
-
The manufacturer of the X-15 steel-belted radial truck tire claims that the mean mileage the tire can be driven before the tread wears out is 60,000 miles. Assume the mileage wear follows the normal...
-
Based on the design, briefly discuss the data collection procedures to be used. Be sure to include the area rea of focus and targeted sample as part of these procedures. Develop a hypothetical...
-
DeLong Corporation was organized on January 1, 2014. It is authorized to issue 10,000 shares of 8%, $100 par value preferred stock, and 500,000 shares of no-par common stock with a stated value of $2...
-
Describe persistence design under NoSQL technologies.? What is NoSQL polyglot persistence?
-
A 1.000-g sample of a liquid that contains only carbon and hydrogen burns in oxygen to produce 1.284 g of H 2 O. (a) What are the mass percents of the elements present in this sample? (b) What is the...
-
Succinic acid, an intermediate in the metabolism of certain foods, has a molecular mass of 118.1 g/mole. A 1.926 g sample of succinic acid (H x Suc) reacts with exactly 1.25 g of NaOH according to...
-
Why does the government mandate individuals to purchase their own insurance income casessuch as automobile liability insurancebut directly provide insurance to people in other situationssuch as...
-
A Difficult Negative MessageTaking Away Extra Pay Your organization employs far more part-time than fulltime workers. Part-timers are paid hourly, up to 15 hours per week, and full-timers are...
-
Write a C++ program that ask the user to Enter 3 characters. If they are all small letter, the program output the message "all small" and output the sum of their ascii codes. If the first and the...
-
Convert the followings from one base to another (you must show the steps as done in classes; two (2) points will be deducted if you don't). 1.a Unsigned 10111012 to a decimal number. Answer: 1.b...
-
6. Write a Verilog program to design an N-bit rotator that has the following capabilities: (a) parallel load data capability (has priority over rotate) (17 points) (b) rotate right and left by 1-bit...
-
Consider the tables provided below and answer the following SQL questions: Table 1: Artist Table 2: Track (Traid - means TrackID) traid title 1 Great Big Words 1 This Pretty Planet 2 Cat's in the...
-
Madison and his adult son lived in a house owned by Madison. At the request of the son, Marshall painted the house. Madison did not authorize the work, but he knew that it was being done and raised...
-
CdF2 (s) Cd+ (aq) + 2 F- (aq) 1. A saturated solution of CdF2 is prepared. The equilibrium in the solution is represented above. In the solution [Cd+] eq = 0.0585 M and [F-] eq = 0.117 M. a....
-
The tetrapeptide Val-Lys-Ala-Phe is cleaved into two fragments upon treatment with trypsin. Identify the sequence of a tetrapeptide that will produce the same two fragments when treated with...
-
Consider the structure of the following cyclic octapeptide. Would cleavage of this peptide with trypsin produce different fragments than cleavage with chymotrypsin? Explain. Phe Arg Arg Phe Phe Arg...
-
Draw all of the steps and reagents necessary to prepare each of the following dipeptides from their corresponding amino acids. (a) Trp-Met (b) Ala-Ile (c) Leu-Val
-
First,Use data to compute the company's pro forma income statement and balance sheet accounts, assuming a 40% tax rate. Sales $46,298,115.00 Retained earnings $12,717,254.00 Other expenses $...
-
When Patey Pontoons Issued 6% bonds on January 1, 2024, with a face amount of $600,000, the market yield for bonds of similar risk and maturity was 10%. The bonds mature December 31, 2027 (4 years)....
-
1. From your chief executive: Whats this I hear about talent management? Is it simply new wine in old bottles as so many innovative HR practices seem to be? Is it for us and if so, why and how? How...

Study smarter with the SolutionInn App


