Assign an oxidation state to each atom in the amino acid glycine: - H-N-C-C T
Question:
Assign an oxidation state to each atom in the amino acid glycine:
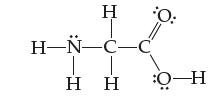
Transcribed Image Text:
Η Η- H-N-C-C T Η Η O-H 0:
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (4 reviews)
The following table shows the oxidation states of each atom in the amino acid glycine Atom Oxidation state H 1 C carbonyl carbon 1 C alpha carbon 1 N ...View the full answer

Answered By

Nyron Beeput
I am an active educator and professional tutor with substantial experience in Biology and General Science. The past two years I have been tutoring online intensively with high school and college students. I have been teaching for four years and this experience has helped me to hone skills such as patience, dedication and flexibility. I work at the pace of my students and ensure that they understand.
My method of using real life examples that my students can relate to has helped them grasp concepts more readily. I also help students learn how to apply their knowledge and they appreciate that very much.
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Introductory Chemistry Atoms First
ISBN: 9780321927118
5th Edition
Authors: Steve Russo And Michael Silver
Question Posted:
Students also viewed these Sciences questions
-
In carbon dioxide, CO 2 , (a) Assign an oxidation state to each atom in the molecule. (b) How many electrons does the C atom own by oxidation-state electron bookkeeping? (c) How many more or fewer...
-
In chloroform, CHCl 3 , (a) Assign an oxidation state to each atom in the molecule. (b) How many electrons does the C atom own by oxidation-state electron bookkeeping? (c) How many more or fewer...
-
In methane, CH 4 , (a) Assign an oxidation state to each atom in the molecule. (b) How many electrons does the C atom own by oxidation-state electron bookkeeping? (c) How many more or fewer valence...
-
Problem 5-47 Amortizing Loans and Inflation (LO3) Suppose you take out a $108,000, 20-year mortgage loan to buy a condo. The interest rate on the loan is 5%. To keep things simple, we will assume you...
-
The comparative balance sheet of Putnam Cycle Co. at December 31, 2010 and 2009, is as follows: The noncurrent asset, noncurrent liability, and stockholders equity accounts for 2010 are as follows:...
-
Dodge City Realty acts as an agent in buying, selling, renting, and managing real estate. The unadjusted trial balance on July 31, 2010, is shown below. The following business transactions were...
-
If a \({ }_{92}^{238} \mathrm{U}\) nucleus is struck by a neutron, it may absorb the neutron. The resulting nucleus then rapidly undergoes betaminus decay. The daughter nucleus of that decay is A....
-
Tar Heel Corporation provides the following information at the end of 2015. Salaries payable to workers at the end of the year ......... $ 3,300 Advertising expense for the year ...............10,400...
-
Explain the role of the following types of data sources in the budget development process. a. Competitor research b. Customer or supplier research c. Declared commitments in areas of operation d....
-
Identify the oxidizing agent and reducing agent in the reaction IO + 71 + 8 H412 + 4 HO
-
When you turn on an electrical appliance, are you consuming electrons?
-
Graph the function. y = 1 + x - 1
-
Characteristics of Competent Communicators . 1. SELF-AWARENESS Good communicators are aware of their own behavior and its effects on others - self-monitoring People who are high self-monitors pay...
-
I must make a storyboard of two rival companies such as sony and nintedo thinking of an announcement by Sony that the Playstation makes to surpass Nintendo that makes nintendo switch consoles for...
-
XYZ Corporation, a multinational manufacturing company, is considering a significant investment in a new production facility. The capital budget for this project involves substantial financial...
-
FISH ANATOMY AND NECROPSY Draw the external and internal anatomy of your specimen and identify each part correctly. You may use extra sheet if necessary. -External Anatomy of Fish -Internal Anatomy...
-
What is the gross yearly income? 2. What is the gross monthly income using this pay rate?
-
Darrell is an employee of Whitneys. During the current year, Darrells salary is Communication Skills $112,000. Whitneys net self-employment income is also $112,000. Calculate the Social Security and...
-
A survey of 70 college freshmen asked whether students planned to take biology, chemistry, or physics during their first year. Use the diagram to answer each question. How many of the surveyed...
-
A current source in a linear circuit has i s = 15 cos (25 t + 25) A (a) What is the amplitude of the current? (b) What is the angular frequency? (c) Find the frequency of the current. (d) Calculate...
-
Given v 1 = 45 sin(t + 30) V and v 2 = 50 cos(t 30) V, determine the phase angle between the two sinusoids and which one lags the other.
-
Transform the following sinusoids to phasors: (a) 20 cos(4t + 135) (b) 8 sin(20t + 30) (c) 20 cos (2t) + 15 sin (2t)
-
You are the owner of a factory located in a hot tropical climate. The monthly production of the factory is $ 1 0 0 , 0 0 0 ?except during June ? ?September, when it falls to $ 8 0 , 0 0 0 ?due to the...
-
Suppose you buy a $ 7 0 0 , 0 0 0 house with ' no money down' with a 3 0 year, fixed - rate 7 % mortgage paid month starting TODAY! After 3 0 years you've finally paid off your mortgage....YEAH How...
-
You want to save $600,000 dollars for retirement in your 401K. Over the next 30 years, How much must you deposit weekly if the annuity is paying 7.35%? What will be your total payments into the...

Study smarter with the SolutionInn App


