Complete the table: Solute Solute mass (g) NaBr 3.96 Ba(OH)2 2.58 (NH4)2SO4 8.65 NH4Cl Solution Molarity volume
Question:
Complete the table:
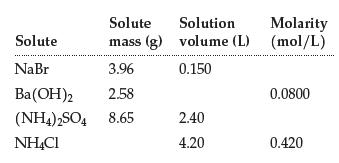
Transcribed Image Text:
Solute Solute mass (g) NaBr 3.96 Ba(OH)2 2.58 (NH4)2SO4 8.65 NH4Cl Solution Molarity volume (L) (mol/L) 0.150 2.40 4.20 0.0800 0.420
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
Molarity M mass g 1 molar mass gmol Liter Molarity M NaBr 396 ...View the full answer

Answered By

Irfan Ali
I have a first class Accounting and Finance degree from a top university in the World. With 5+ years experience which spans mainly from the not for profit sector, I also have vast experience in preparing a full set of accounts for start-ups and small and medium-sized businesses. My name is Irfan Ali and I am seeking a wide range of opportunities ranging from bookkeeping, tax planning, business analysis, Content Writing, Statistic, Research Writing, financial accounting, and reporting.
4.70+
249+ Reviews
530+ Question Solved
Related Book For 

Introductory Chemistry Atoms First
ISBN: 9780321927118
5th Edition
Authors: Steve Russo And Michael Silver
Question Posted:
Students also viewed these Sciences questions
-
For an ideal gas, the decrease in internal energy of 1.4 kgm is -343KJ when the volume increases from 0.043 cu. m. to 0.13 from 0.07 bar to 0.02 bar; c, = 0.72 cu. m. and the pressure decreases kgm-K...
-
Complete the table below for contribution margin per unit, total contribution margin , and contribution margin ratio: A Number of units 1,720 units 14,920 units 4,620 units Sales price per unit $...
-
Problem 13-1A Calculation and analysis of trend percents LO A1, P1 Selected comparative financial statements of Haroun Company follow. HAROUN COMPANY Comparative Income Statements For Years Ended...
-
Why will a reduction in the real interest rate increase investment spending, other things equal?
-
A machine is purchased January 1 at a cost of $58,000. It is expected to produce 110,000 units and have a salvage value of $3,000 at the end of its useful life. Units produced are as follows: Year 1...
-
What is the ultimate goal of a lean system? What are the supporting goals? What are the building blocks?
-
Larry Riley formed a rental agreement for a self-storage unit with Iron Gate Self Storage in December 2003. The agreement stated that a maximum of \($5,000\) of personal property could be stored in...
-
Three different plans for financing a $40,000,000 corporation are under consideration by its organizers, under each of the following plans, the securities will be issued at their par of face amount,...
-
What are the drawbacks (disadvantages) of the relational database management system and of the object-oriented database system? What hardware or software support has been implemented for the...
-
Calculate the number of moles of each ion present in 2.00 10 2 cm 3 of (a) 0.200 M NaCl, (b) 0.350 M K 3 PO 4 , (c) 1.44 M Al(NO 3 ) 3 .
-
How would you prepare 250.0 mL of a 0.350 M NaOH solution from a 6.00 M NaOH stock solution?
-
Since the computer had done most of his calculations in the past, Justin decided to check to see if the computer was the source of the problem. He gathered information to conduct a manual calculation...
-
A taxpayer has a mortgage with a total value of $1,500,000 and a home equity loan of $250,000, both of which were finalized in TY2016. The total interest paid during the year was $54,000 for the...
-
What is the focus when analyzing the operating cycle of a business?
-
What root operation is angioplasty classified to?
-
Kevin borrowed a 25-year mortgage loan of $8,000,000 from Hang Seng Bank (HSB) to purchase a flat in North Point 6 years ago. The interest rate was made at "Prime rate minus 2.7%". The loan was a...
-
The company's adjusted trial balance includes the following accounts balances: Cash, $ 1 5 , 0 0 0 ; Equipment, $ 8 5 , 0 0 0 ; Accumulated Depreciation, $ 2 5 , 0 0 0 ; Accounts Payable, $ 1 0 , 0 0...
-
A survey conducted by the Northwestern University Lindquist-Endicott Report asked 320 companies about the procedures they use in hiring. Only 54% of the responding companies review the applicant's...
-
The process of collaborative goal setting by a manager and subordinate, the extent to which goals are accomplished is a major factor in evaluating and rewarding the subordinate's performance. It is...
-
The pin support is made from a steel rod and has a diameter of 20 mm. Determine the stress components at points C and D and represent the results on a volume element located at each of these points....
-
The pin support is made from a steel rod and has a diameter of 20 mm. Determine the stress components at points A and B and represent the results on a volume element located at each of these points....
-
The uniform sign has a weight of 1500 lb and is supported by the pipe AB, which has an inner radius of 2.75 in. and an outer radius of 3.00 in. If the face of the sign is subjected to a uniform wind...
-
Simplify. 2 x-4)2x +0x - 26x - 24
-
On January 1 , 2 0 X 2 P pays $ 1 0 0 million to acquire 5 0 , 0 0 0 of the common voting shares of S , which gives P a 2 5 % ownership interest in S . The January 1 , 2 0 X 2 book value of S ' s...
-
Shaw Company has the following account balances: Receivables $ 1 0 0 , 0 0 0 Inventory 1 5 0 , 0 0 0 Land 1 0 0 , 0 0 0 Building net 2 5 0 , 0 0 0 Liabilities 1 0 0 , 0 0 0 Common stock 1 0 0 , 0 0 0...

Study smarter with the SolutionInn App


