Convert this excited boron, B, atom to (a) An excited state of lower energy (b) The
Question:
Convert this excited boron, B, atom to
(a) An excited state of lower energy
(b) The ground state:
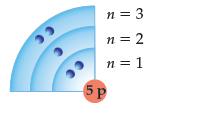
Transcribed Image Text:
33 СС i сл р n = 3 n=2 n = 1
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 33% (3 reviews)
a An excited state of lower energy To convert the excited boron atom in the image to an excited state of lower energy we need to move one of the elect...View the full answer

Answered By

HARSH RANJAN
Taken classes at college to graduates, Also worked as an expert to a freelancer online question-solving portal for more than 8 months with an average rating greater than 4.2 out of 5.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Introductory Chemistry Atoms First
ISBN: 9780321927118
5th Edition
Authors: Steve Russo And Michael Silver
Question Posted:
Students also viewed these Sciences questions
-
An atom in an excited state will on the average undergo a transition to a state of lower energy in about 10 -8 seconds. If the electron in a doubly ionized lithium atom (Li +2 , which is...
-
A transition of particular importance in 02 gives rise to the 'Schumann-Rung c band' in the ultraviolet region. The wave numbers (in cm-1) of transitions from the ground state to the vibrational...
-
An electron in a hydrogen atom is excited from the ground state to the n = 4 state. Comment on the correctness of the following statements (true or false). (a) n = 4 is the first excited state. (b)...
-
What does it mean that flame is non-luminous it is yellow it is invisible It has a very bright blue inner cone it is dark
-
What is a standard? Give some examples such as those appearing in the chapter introduction.
-
Assume that you just completed a December weekend vacation to a casino within the United States. During your trip you won $ 10,000 gambling. When the casino exchanged your chips for cash they did not...
-
A university pumps its water from wells located on campus. The falling water table has caused pumping costs to increase, the quantity of water available to decrease, and the quality of water to...
-
Wyco Company manufactures toasters. For the first 8 months of 2011, the company reported the following operating results while operating at 75% of plant capacity. Sales (400,000 units) ......
-
Consider two oppositely charged atoms with charges of +1 and -1 units, respectively. The two atoms interact with each other through electrostatic (Coulombic) and van der Waals forces, and reach an...
-
According to Bohrs model, is energy absorbed or released when an electron moves to a shell of lower n?
-
Would moving an electron farther from an atoms nucleus give off light energy or require the absorption of light energy? Explain your answer.
-
Sardano and Sons is a large, publicly held company that is considering leasing a warehouse. One of the companys divisions specializes in manufacturing steel and this particular warehouse is the only...
-
Explain the difference between a cash dividend, a stock dividend, a special dividend, and a stock buyback.
-
What is the catering theory of dividends and how would it influence a manager looking to improve the value of her firm through dividend policy? What does the empirical evidence say about the catering...
-
Explain the role of agency conflicts between managers, shareholders, and bondholders in corporate payout policy.
-
Managerial Motives for Takeovers Outline the managerial motives for takeovers.
-
Explain the importance of taxes in dividend policy. What are the real world factors that would encourage firms to follow a high dividend policy?
-
Outdoor World Inc. (OWI) is a sporting goods retailer that specializes in bicycles, running shoes, and related clothing. The firm has become successful by careful attention to trends in cycling,...
-
Which of the following statements is false? a. Capital leases are not commonly reported in a Capital Projects Fund. b. A governmental entity may report a Capital Project Fund in one year but not the...
-
There are more electrons in the n = 4 shell than for the n = 3 shell in krypton. However, the peak in the radial distribution in Figure 21.6 is smaller for the n = 4 shell than for the n = 3 shell....
-
How is the effective nuclear charge related to the size of the basis set in a Hartree Fock calculation?
-
Using the result of Problem P20.12, calculate the probability of finding the electron in the 1s state outside a sphere of radius 0.75a 0 , 2.5a 0 , and 4.5a 0 .
-
Discuss the importance of a comprehensive Onboarding process as an individual transition from job candidate to a new employee.
-
Trace the following logic and answer the questions that follow. Logic W = 0 X = 3 Y = 5 Z = 5 Counter = 1 Do Until X > 7 If Y2 and Y < 6 Y = Y + 2 Z = Z +5 For Counter = 1 to 5 W Counter + 1 = Next...
-
Consider the following two classes Node and CircularlyLinkedList: private class Node { private int element; private Node next; public Node (int e, Node n) { element = e; next = n; } public int...

Study smarter with the SolutionInn App


