Draw structural formulas for the hydrocarbons in Practice Problem 17.1, showing all carbons, hydrogens, and bonds. Data
Question:
Draw structural formulas for the hydrocarbons in Practice Problem 17.1, showing all carbons, hydrogens, and bonds.
Data from Practice Problem 17.1
Give the molecular formula for each hydrocarbon molecule:
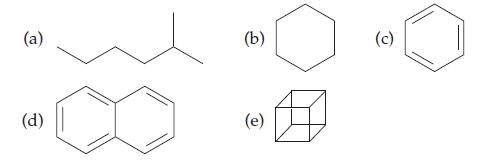
Transcribed Image Text:
(a) (d) (b) (e) (c)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
a H H H H H CH H H HS HC CH H b HC C...View the full answer

Answered By

Fahmin Arakkal
Tutoring and Contributing expert question and answers to teachers and students.
Primarily oversees the Heat and Mass Transfer contents presented on websites and blogs.
Responsible for Creating, Editing, Updating all contents related Chemical Engineering in
latex language
4.40+
8+ Reviews
22+ Question Solved
Related Book For 

Introductory Chemistry Atoms First
ISBN: 9780321927118
5th Edition
Authors: Steve Russo And Michael Silver
Question Posted:
Students also viewed these Sciences questions
-
Draw structural formulas for the isomers of ethylmethylbenzene.
-
Draw structural formulas for all possible isomers having the following molecular formulas: a. C3H8 b. C3H7F c. C2H2Br2 d. C3H6 e. C4H9Cl f. C3H6Cl2 g. C3H8S h. C2H4F2
-
Draw structural formulas for the five isomers of C6H14. As you write them out, try to be systematic, starting with a consecutive chain of six carbon atoms.
-
8. Describe each of the following types of isomerism (i) geometrical isomerism (2 marks) (ii) solavate isomerism (2 marks) (iii) coordination isomerism (2 amrks)
-
Suppose demand and supply are given by Qd = 50 P and Qs = P 10. a. What are the equilibrium quantity and price in this market? b. Determine the quantity demanded, the quantity supplied, and the...
-
The Vaderson Forecasting Associates sells a broad range of economic forecasting services to businesses and government agencies. One of its primary products is the Vaderson Exchange Rate Seer, a model...
-
An experiment in chemistry is repeated, and the times it takes for a reaction to occur are recorded. State whether the data described are discrete or continuous and explain why?
-
Norma Smith is the controller of Baylor Corporation and is responsible for the preparation of the year-end financial statements. The following transactions occurred during the year. (a) On December...
-
What is a way that compensation professionals can help leverage the value of human capital?
-
Consider the branched hydrocarbon: (a) What is the length of the main chain? (b) What is the length of the branch? H_c-c-c-C-C-H | | T I H-C-H HC-H HC-H T
-
The following statement is true about carbon: (a) Because a carbon atom has four valence electrons, it typically forms four bonds to other atoms. (b) Carbon atoms can catenate, which means they can...
-
A candy maker mixes chocolate, milk, and mint extract to produce three kinds of candy (I, II, and III) with the following proportions: I: \(7 \mathrm{lb}\) chocolate, 5 gal milk, \(1 \mathrm{oz}\)...
-
Mary Rossi, a student at State College, has a balance of $380 on her retail charge card; if the store levies a finance charge of 21% per year, how much monthly interest will be added to her account?
-
Carla Esposito has an overdraft protection line. Assume that her October 2010 statement showed a latest (new) balance of $862. If the line had a minimum monthly payment requirement of 5% of the...
-
Michael Sutton is trying to decide whether to apply for a credit card or a debit card. He has $7,500 in a savings account at the bank and spends his money frugally. What advice would you have for...
-
Dean Rickert plans to borrow $8,000 for 5 years. The loan will be repaid with a single payment after 5 years, and the interest on the loan will be computed using the simple interest method at an...
-
Ramon and Maria Lopez are a newly married couple in their mid-20s. Ramon is a senior at a state university and expects to graduate in the summer of 2011. Maria graduated last spring with a degree in...
-
VBK, Inc., applies overhead costs to its products using direct labor costs as its cost driver. For the most recent year, its budgeted overhead costs were $525,000, and its budgeted direct labor costs...
-
Multiple Choice Questions: 1. The largest component of aggregate demand is? a. Government purchases. b. Net exports. c. Consumption. d. Investment. 2. A reduction in personal income taxes, other...
-
An altimeter is a device used to measure altitude. Most altimeters are based on measurements of air pressure. Explain how they work and estimate how much the pressure changes for a 100-m change in...
-
The water in the pipes of a typical house is at a gauge pressure of 3.0 times atmospheric pressure. What is the absolute pressure in pascals and in units of pounds per square inch?
-
In preparation for a bike tour of several countries, you ship your bicycle by air freight to Europe. When you arrive, you find that the tires of your bicycle have both blown out. Explain why that...
-
2.9 A closed-cycle gas turbine is to be used in conjunction with a gas-cooled nuclear reactor. The working fluid is helium (cp = 5.19 kJ/kg K and y = 1.66). The layout of the plant consists of...
-
6.32 A material with a yield strength of Sy 350 MPa is subjected to the stress state shown in Sketch c. What is the factor of safety based on the maximum shear stress and distortion energy theories?...
-
The shaft shown in the figure has a diameter of 23.3 mm and is supported by smooth journal bearings at A and B. Due to the transmission of power to and from the shaft, the belts on the pulleys are...

Study smarter with the SolutionInn App


