Fill in this table: Name Sodium phosphate Iron(II) carbonate Mercury(II) chloride Formula Ba(CH3CO)2 (NH4)2S Co(OH)2 HgCl Soluble
Question:
Fill in this table:
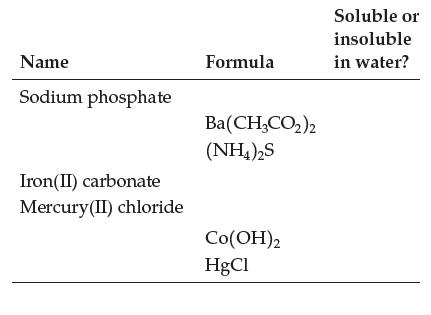
Transcribed Image Text:
Name Sodium phosphate Iron(II) carbonate Mercury(II) chloride Formula Ba(CH3CO₂)2 (NH4)2S Co(OH)2 HgCl Soluble or insoluble in water?
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
Compound Formula Soluble in water Sodium phosphate Na3PO4 Yes Barium acetate BaCH3COO2 Yes Ammonium ...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Introductory Chemistry Atoms First
ISBN: 9780321927118
5th Edition
Authors: Steve Russo And Michael Silver
Question Posted:
Students also viewed these Sciences questions
-
Fill in this table with one of the following statements: no error-the process is good, no error-the process is bad, type I or alpha error, type II or beta error. In reality, if we looked at...
-
The VLOOKUP function in Excel is a powerful tool that manystudents are expected to master for business-related careers. Asits name implies, VLOOKUP relies on a "lookup" table. In Excel, alookup table...
-
Assess the four reasons why the selection of the FOB point is important to the construction company as a purchaser. Provide relevant examples to support your answer.
-
Find or evaluate the integral by completing the square. 3 L= dx x + 4x + 8
-
Borysko Company makes and sells products with variable costs of $47 each. Borysko incurs annual fixed costs of $22,400. The current sales price is $63. Required The following requirements are...
-
Akron Manufacturing Company manufactures a cement sealing compound called Ultra-Seal. The process requires that the product pass through three departments. In Dept. 1, all materials are put into...
-
How does a signal from a motor neuron result in the contraction of a muscle?
-
Griffon Corp. makes small plastic dog toys with the following material and labor standards: During October, 60,000 pounds of material were acquired on account at $4.15 per pound. During October,...
-
4. **Boomerang. If a boomerang is thrown correctly, it returns to the thrower. Most boomerangs consist of two arms separated by about 90 but the four-blade boomerang (see figure) has the same physics...
-
Write a net ionic equation for the precipitation reaction, if any, that occurs when aqueous solutions of the following ionic compounds are mixed: (a) Bi(NO 3 ) 3 and NaOH (b) K 2 SO 4 and SrI 2 (c)...
-
Draw a beaker that shows the result of dissolving methanol, CH 3 O H , in water and a second beaker that shows the result of dissolving sodium phosphate in water. Indicate all species present in...
-
Do mobile devices allow you to show more information on a screen, or less?
-
Explain the process of an IPO.
-
What are the sources and uses of funds in insurance?
-
Discuss the main characteristics of common stock.
-
Distinguish between takaful insurance and conventional insurance.
-
What are the different types of MBS?
-
A democracy is based on the idea that for many purposes, the best decision-making framework is to resolve difficult issues by following the wishes of the representatives of majority opinion....
-
Burberrys competitive advantage is through its differentiation strategy. What risk should Burberry remain aware of?
-
Seawater freezes at a lower temperature than pure water because of the salts dissolved in it. How does the boiling point of seawater compare with that of pure water?
-
What is the easiest way to distinguish between a solution that contains Cu 2+ ions from one that contains Ca 2+ ions?
-
You have a solution that contains Ca 2+ ions and another that contains Na + ions. How would adding a solution that contains CO 2- 3 ionss enable you to tell which is which?
-
#10. Bay Pine Medical Center estimates that a capitated population of 50,000 would have the following base case utilization and total cost characteristics: In addition to medical costs, Bay Pines...
-
Consider this. A man dining in a restaurant experiences a heart attack. The staff immediately calls 911 but ignores a nearby emergency defibrillator. They remain with the guest until the ambulance...
-
Access the IASB Codification and answer the following: According to guidance in the IASB Codification, IASB 2 - Inventories, how should one account inventory costs using their cost formulas? How is...

Study smarter with the SolutionInn App


