In which period and group in the periodic table are these atoms found? (a) 1s2s2p (b) 1s2s2p63s
Question:
In which period and group in the periodic table are these atoms found?
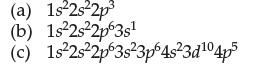
Transcribed Image Text:
(a) 1s²2s²2p³ (b) 1s²2s²2p63s¹ (c) 1s²2s²2p 3s²3p64s²3d¹04p5
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (4 reviews)
a The electron configuration 1s2 2s22p3 belongs to an element in pe...View the full answer

Answered By

Nazrin Ziad
I am a post graduate in Zoology with specialization in Entomology.I also have a Bachelor degree in Education.I posess more than 10 years of teaching as well as tutoring experience.I have done a project on histopathological analysis on alcohol treated liver of Albino Mice.
I can deal with every field under Biology from basic to advanced level.I can also guide you for your project works related to biological subjects other than tutoring.You can also seek my help for cracking competitive exams with biology as one of the subjects.
3.30+
2+ Reviews
10+ Question Solved
Related Book For 

Introductory Chemistry Atoms First
ISBN: 9780321927118
5th Edition
Authors: Steve Russo And Michael Silver
Question Posted:
Students also viewed these Sciences questions
-
Which of the first 10 elements in the periodic table are found as free elements in the Earths crust? Which elements in this group occur in the Earths crust only as part of a chemical compound?
-
Consider the following information about the newly discovered element, vulcium, whose symbol is Vu. Vulcium is a solid at room temperature. It is easily cut by a penknife to reveal a shiny surface...
-
Elements in group 7A in the periodic table are the halogens; elements in group 6A are called the chalcogens. (a) What is the most common oxidation state of the chalcogens compared to the halogens?...
-
What is the common name for the following structure? CH3 CH3 -C-Br 1 CH3 Isobutyl bromide Osec-Butyl bromide O Neobutyl bromide O Isopropyl methyl bromide t-Butyl bromide
-
General Mills is a company that uses process costing extensively. Go to the companion Web site at www.cengage.com/accounting/vanderbeck and click on the file for General Mills. Required: Answer the...
-
As learners, what should you demonstrate to help yourself and our educators make the education successful during a time of crisis? In this time of crisis, our regular physical attendance in our...
-
In the spring of 1999, Source Associates, Inc. (Source), and Conrad A. Mamajek, Inc. (CAM), entered into a joint venture to act as a middleman for the sale of polymers manufactured by Mitsui...
-
The Cutting Department of Cassel Company has the following production and cost data for July. Materials are entered at the beginning of the process. Conversion costs are incurred uniformly during the...
-
the tuition fee for maths tuition for john is expected to be 6500 per year if he attend tuition for 4 year starting from 4 years from now how much does he have to have in her account which yields 6...
-
Repeat Problem 4.86, but use the noble gas abbreviated form this time. Data from Problem 4-86 Write the electron configuration for the following elements without using the noble gas abbreviated form....
-
Write the electron configuration for the following elements without using the noble gas abbreviated form. (a) Ba (b) W (c) Pb (d) Pr (e) Pa
-
Cell motility has been described as being like the motion of tank treads. At the leading edge, actin filaments form rapidly into bundles and networks that make protrusions and move the cell forward....
-
What is the average cost of bankruptcy for a firm in the United States? How does this compare to other countries? Why do bankruptcy costs differ both within a country and between countries?
-
Why do you think Power Assets really reduced their payables period? Is their explanation that it would control costs and optimize cash flow sensible? Will there be any direct or indirect cash...
-
Behavioural Finance In recent years, a new interpretation of market behaviour has emerged. Explain this theory and review what it says about Shleifers three conditions of market efficiency. Compare...
-
Survivor NV, an all-equity firm, has three shares outstanding. Yesterday, the firms assets consisted of 5 ounces of platinum, currently worth 1,000 per ounce. Today, the company issued Ms Wu a...
-
Efficient Capital Markets What are the three conditions under which Fama (1970) argues markets may be efficient? Describe these in detail and provide a practical example illustrating each condition...
-
Can the marketing department be both a revenue center and a cost center? Explain.
-
Baxter, Inc., owns 90 percent of Wisconsin, Inc., and 20 percent of Cleveland Company. Wisconsin, in turn, holds 60 percent of Clevelands outstanding stock. No excess amortization resulted from these...
-
Identify the molecular orbitals for F 2 in the images shown here in terms of the two designations discussed in Section 23.7. The molecular axis is the z axis, and the y axis is tilted slightly out of...
-
Why are the magnitudes of the coefficients c a and c b in the H 2 + wave functions g and u equal?
-
Predict the bond order in the following species: a. N + 2 b. Li + 2 c. O 2 d. H 2 e. C + 2
-
find at least two recent articles that describes new or developing technology that will have significant influence on supply chain management within the next 5-6 years. You will need to add your...
-
Watch the film Mega Mall shown in class (retail development) (link provided on wk7, Canvas) and answer the following questions: Why was thePalisades Center, at Clark's town a controversial project?...
-
Forecasting is essential to improving a hotel's future performance. If you are the manager of a hotel: What types of forecasts will be needed? What demand forecasting methods would you use to...

Study smarter with the SolutionInn App


