Of the following transitions in the Bohr hydrogen atom, which transition results in the emission of photons
Question:
Of the following transitions in the Bohr hydrogen atom, which transition results in the emission of photons with the shortest wavelength?
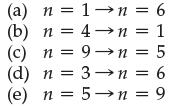
Transcribed Image Text:
n = 6 (a) (b) (c) n=1 n =4 n = 1 = 5 n=9→n (d) n = 3→n = 6 (e) n = 5→n = 9
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (4 reviews)
The transition that results in the emission of photons with the shortest wavelength in the Bohr hydr...View the full answer

Answered By

Krishnavendra Y
I am a self motivated financial professional knowledgeable in; preparation of financial reports, reconciling and managing accounts, maintaining cash flows, budgets, among other financial reports. I possess strong analytical skills with high attention to detail and accuracy. I am able to act quickly and effectively when dealing with challenging situations. I have the ability to form positive relationships with colleagues and I believe that team work is great key to performance. I always deliver quality, detailed, original (0% plagiarism), well-researched and critically analyzed papers.
5.00+
4+ Reviews
10+ Question Solved
Related Book For 

Introductory Chemistry Atoms First
ISBN: 9780321927118
5th Edition
Authors: Steve Russo And Michael Silver
Question Posted:
Students also viewed these Sciences questions
-
Consider only transitions involving the n = 1 through n = 4 energy levels for the hydrogen atom (see Figures 6.7 and 6.10). (a) How many emission lines are possible, considering only the four quantum...
-
The cosmic dawn that preceded the epoch of reionization can be probed by low frequency CMB observations using a special radio hyperfine line emitted and absorbed by hydrogen atoms. This line is...
-
Consider only transitions involving the n = 1 through n = 5 energy levels for the H atom (see Figures 6.7 and 6.10). (a) How many emission lines are possible, considering only the five quantum...
-
Name: PN 200 Fundamentals of Nursing II Medication Error Prevention-OTC's Date: Jeff Voss, a 29-year-old graduate student is at the student health center for a physical examination required before...
-
What are six principles of good budgeting?
-
In the Seafood Companys packaging department, conversion costs are added evenly during the process, and direct materials are added at the end of the process. Spoiled units are detected upon...
-
James Lewis, a resident of Kentucky, sustained an injury while operating a Caterpillar bulldozer. He filed suit against Caterpillar, a company incorporated in Delaware but with its principal place of...
-
Morse Company manufactures basketballs. Materials are added at the beginning of the production process and conversion costs are incurred uniformly. Production and cost data for the month of July 2012...
-
Consider the following logarithmic equation. log3(x) log.x 8- log(x) log(x) = 1. (i) Find the value(s) of a satisfying in the equation. (ii) Determine for what values of the logarithmic expression on...
-
When a hydrogen atom is excited in a flame, a line of blue-violet light is emitted. This happens when an electron makes a transition between the n = 5 and the n = 2 orbit of the atom. If the energy...
-
In which group and period would you expect to find the MOST nonmetallic element?
-
An increase in the minimum wage reduces the total amount paid to the affected workers if the price elasticity of _________ is _________ than one. a. supply; greater b. supply; less c. demand; greater...
-
What is the approach by which we can effectively interpret a firms financial ratios?
-
Where can we find financial ratios for different companies or for peer groups?
-
How is a companys return on equity related to the firms debt ratio?
-
SCOP TI is considering introducing a variation of its current breakfast tea, Earl Grey 1336. The new tea bags will be similar to the old with the exception that it will contain small caramel-flavored...
-
Why should you be careful when comparing a firm with industry norms?
-
Explain the three types of management compensation.
-
Select a mass spectrometric technique with the highest mass resolution for identifying an unknown compound being eluted from a liquid chromatography column
-
Why isnt the motion of a human being described by the Schrdinger equation rather than Newtons second law if every atom in our body is described by quantum mechanics?
-
Explain the following statement: If h = 0, it would be possible to measure the position and momentum of a particle exactly and simultaneously.
-
Why is (p 2 ) rather than p used to calculate the relative uncertainty for the particle in the box?
-
Instructions - President Theater 38518DL Questions will be taken in the Student Lounge Discussion Group 1Enter Your Name Right Here, in Cell B3 - NOW This will put your name on all the pages S22 Save...
-
(b) Based on the table below, you invested 40% on Stock A and B and 20% on Stock C. Calculate the expected return on this portfolio. Year Expected rate of returns Stock A Stock B Stock C 1 0.18 0.13...
-
"How do technological advancements, such as social media platforms, big data analytics, and algorithmic recommendation systems, shape online influence dynamics, opinion polarization, and echo chamber...

Study smarter with the SolutionInn App


