Redraw each structure to get the electron groups as far apart as possible. What angles did you
Question:
Redraw each structure to get the electron groups as far apart as possible. What angles did you use?
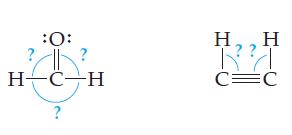
Transcribed Image Text:
:O: ? H CH ? H RA C=C H
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
120 120 H ...View the full answer

Answered By

GERALD KAMAU
non-plagiarism work, timely work and A++ work
4.40+
6+ Reviews
11+ Question Solved
Related Book For 

Introductory Chemistry Atoms First
ISBN: 9780321927118
5th Edition
Authors: Steve Russo And Michael Silver
Question Posted:
Students also viewed these Sciences questions
-
Planning is one of the most important management functions in any business. A front office managers first step in planning should involve determine the departments goals. Planning also includes...
-
THIRD AVENUE SOFTWARE HEALTH-CARE APP PROJECT This case is new for the ninth edition of Information Technology Project Management . The case provides an opportunity to apply agile and Scrum...
-
Managing Scope Changes Case Study Scope changes on a project can occur regardless of how well the project is planned or executed. Scope changes can be the result of something that was omitted during...
-
Fill in the missing mass number and atomic number for each of these particles or types of radiation. alpha (?) He beta (?) e gamma y neutron n
-
Do you agree that the company is guilty of illegal actions? Why or why not?
-
Musical Corporation acquires 80 percent of Dustin Corporations common shares on January 1, 20X2. On January 2, 20X2, Dustin acquires 60 percent of Rustic Corporations common stock. Information on...
-
The Anchor Glass Container Corporation and its parent company, Consumers Packaging, Inc. (CPI), entered into a series of agreements with Encore Glass, Inc., to supply glass containers of a specific...
-
Presented below are selected transactions at Tomas Company for 2014. Jan. 1 Retired a piece of machinery that was purchased on January 1, 2004. The machine cost $58,000 on that date. It had a useful...
-
Matt has worked as an electrician for 24 years with the same company and is about to retire. He has a final-earnings pension plan. His pension benefit will be 1% per year based on the average of his...
-
Describe the geometry of the electron groups and name the molecular shape resulting from that geometry. Also, draw the molecule, and label the size of all bond angles in your drawing. CO 2 .
-
The diagram below shows how bent water molecules in solid ice orient themselves with respect to each other. Notice the large openings in the lattice of water molecules. (a) Discuss why it takes...
-
Write an algorithm to settle the following question: A bank account starts out with $10,000. Interest is compounded monthly at 6 percent per year (0.5 percent per month). Every month, $500 is...
-
What is a low-end disruption strategy?
-
What are four sets of challenges to sustained competitive advantage outlined in this chapter?
-
What do we mean by generic strategies?
-
How are people relevant to strategy formulation and implementation?
-
How is international expansion related to business and corporate strategy? Identify a firm that may be thinking of expanding into new international markets. Apply the staging element of the strategy...
-
Big Spring Brewery (BSB) makes two specialty beers in its micro-brewery: Big Springs Ale and Dark Springs, BSB's dark beer. Both beers sell for the same price per case in the U.S. market and in the...
-
How does Kant answer Humes bundle theory of self? Do you think he is successful?
-
How many rotational degrees of freedom are there for linear and nonlinear molecules?
-
Assuming 19 F 2 and 35 Cl 2 have the same bond length, which molecule do you expect to have the largest rotational constant?
-
Consider the rotational partition function for a polyatomic molecule. Can you describe the origin of each term in the partition function, and why the partition function involves a product of terms?
-
a. In a college class, the average IQ is 115. Assume that the distribution is normal and that the standard deviation is 15. What percentage of the class has an IQ between 105 and 130? Use a z table....
-
15. A tank contains 500 liters of brine with 250 grams of salt in the solution. Pure water runs into the tank at the rate of 15 liters/minute and the mixture, constantly stirred, is drained out at...
-
Mary purchased a throw rug priced at $150 and marked 10% off, a coffee table priced at $80 and marked 15% off, and a lamp priced at $65. She had a coupon good for an additional 20% off the entire...

Study smarter with the SolutionInn App


