Redraw the shells of Li, Be, B, and C shown in the above drawing to make the
Question:
Redraw the shells of Li, Be, B, and C shown in the above drawing to make the overall size of each atom fit the size trend. In each nucleus, write the total number of protons present. Which atom would be most difficult to ionize?
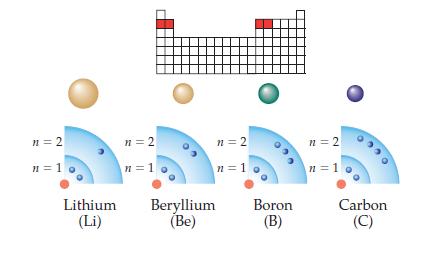
Transcribed Image Text:
n = 2 n=1 Lithium (Li) n=2 n=1 n = 2 n=1 Beryllium (Be) Boron (B) n=2 n=1 Carbon (C)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
Carbon would be the hardest to ionize because its shells are cl...View the full answer

Answered By

Carly Cimino
As a tutor, my focus is to help communicate and break down difficult concepts in a way that allows students greater accessibility and comprehension to their course material. I love helping others develop a sense of personal confidence and curiosity, and I'm looking forward to the chance to interact and work with you professionally and better your academic grades.
4.30+
12+ Reviews
21+ Question Solved
Related Book For 

Introductory Chemistry Atoms First
ISBN: 9780321927118
5th Edition
Authors: Steve Russo And Michael Silver
Question Posted:
Students also viewed these Sciences questions
-
Planning is one of the most important management functions in any business. A front office managers first step in planning should involve determine the departments goals. Planning also includes...
-
Read the case study "Southwest Airlines," found in Part 2 of your textbook. Review the "Guide to Case Analysis" found on pp. CA1 - CA11 of your textbook. (This guide follows the last case in the...
-
Read the case study and answer the question below with a one page response. What does a SWOT analysis reveal about the overall attractiveness of Under Armours situation? Founded in 1996 by former...
-
11) log, 16 4X220 4 ky 13) log, 8+log, (1-4x)=log, 63 15) log, 3x-log, 5=4 12) Evaluate 2 log, 7776-log, 729 14) log, (x-6)+log, (x-5)=1 loge (x-6)+ log 6 (x-5' 16) log 9+ logx = 4
-
What is the major difference between the disposition of units transferred out of a first department in a single-department factory versus a multiple-department factory?
-
Discuss how the inventory process supports the production planning process and the risks to the production process if inventory process control goals are not achieved. Do not limit your discussion to...
-
Jakes Roasted Peanuts, Inc., supplies snack foods. The business experienced the following events. a. Jakes Roasted Peanuts issued common stock after receiving cash from the sole stockholder. b. Cash...
-
Yarnell Electronics sells computer systems to small businesses. Yarnell engaged in the following activities involving notes receivable: a. On November 1, 2011, Yarnell sold a $5,000 system to Ross...
-
A car travelling at 35 mph suddenly collides with a concrete wall. The driver is wearing a seatbelt and is not seriously hurt. The passenger next to the driver does not have their seat belt on. Upon...
-
Why is an electron in a shell that has a low value of n in a more stable arrangement than one in a shell that has a higher value of n?
-
The following statement(s) is true about the periodic table: (a) Elements in the same group have similar valence-shell electron configurations. (b) Elements in the same period have the same value of...
-
Which is more critical to sustaining human life-water or diamonds? Why do firms that provide water to customers generally earn lower economic performance than firms that provide diamonds?
-
____________ refers to the chronic exhaustion from persistent workload, decreased motivation, and apathy toward work.
-
What is the level-of-interactivity test?
-
Review the organizational patterns discussed in this chapter. Considering this information, what preference do you have related to the way you organize information for a presentation?
-
What is a web-wrap agreement?
-
How can a domain name infringe some other persons trademark?
-
Assume that X is a Poisson random variable with = 1.5. Calculate the following probabilities. a. P(X = 1) b. P(X = 2) c. P(X 2)
-
Briefly describe the following types of group life insurance plans: a. Group term life insurance b. Group accidental death and dismemberment insurance (AD&D) c. Group universal life insurance d....
-
The ground electronic state of O 2 is 3 g and the next two highest energy states are 1 g (7918 cm 1 ) and 1 + g (13195 cm 1 ), where the value in parentheses is the energy of the state relative...
-
Determine if the following transitions are allowed or forbidden: a. 3 u 3 g b. 1 + g 1 g c. 3 g 3 g d. 1 g 1 u
-
In a simple model used to analyze UV photoelectron spectra, the orbital energies of the neutral molecule and the cation formed by ejection of an electron are assumed to be the same. In fact, some...
-
A company understated its ending inventory balance by $ 9 , 0 0 0 in 2 0 2 4 . What impact will this error have on total assets and retained earnings in 2 0 2 4 ( ignoring tax effects ) ? What impact...
-
CC Corporation provides you with the following information from its accounting records this year related to income and all book - tax differences. Pre - tax book income: $ 3 1 9 , 0 0 0 GAAP warranty...
-
Based on this information, create and solve a ratio analysis: In their most recent discussion with VP Finance, they were informed that total sales next year ( 2 0 2 2 ) was expected to be $ 1 , 5 0 0...

Study smarter with the SolutionInn App


