The reaction is run at one temperature in flask (a) and at a different temperature in flask
Question:
The reaction
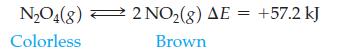
is run at one temperature in flask (a) and at a different temperature in flask (b). Which flask is at the lower temperature, and which is at the higher temperature? Explain your answer.
Transcribed Image Text:
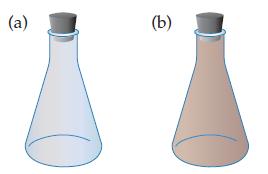
N₂O4(8) 2 NO₂(g) AE = +57.2 kJ Colorless Brown
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
Determining Temperature in Flasks The given information Reaction NO4g 2 NOg H 572 kJ endothermic Fla...View the full answer

Answered By

Somshukla Chakraborty
I have a teaching experience of more than 4 years by now in diverse subjects like History,Geography,Political Science,Sociology,Business Enterprise,Economics,Environmental Management etc.I teach students from classes 9-12 and undergraduate students.I boards I handle are IB,IGCSE, state boards,ICSE, CBSE.I am passionate about teaching.Full satisfaction of the students is my main goal.
I have completed my graduation and master's in history from Jadavpur University Kolkata,India in 2012 and I have completed my B.Ed from the same University in 2013. I have taught in a reputed school of Kolkata (subjects-History,Geography,Civics,Political Science) from 2014-2016.I worked as a guest lecturer of history in a college of Kolkata for 2 years teaching students of 1st ,2nd and 3rd year. I taught Ancient and Modern Indian history there.I have taught in another school in Mohali,Punjab teaching students from classes 9-12.Presently I am working as an online tutor with concept tutors,Bangalore,India(Carve Niche Pvt.Ltd.) for the last 1year and also have been appointed as an online history tutor by Course Hero(California,U.S) and Vidyalai.com(Chennai,India).
4.00+
2+ Reviews
10+ Question Solved
Related Book For 

Introductory Chemistry Atoms First
ISBN: 9780321927118
5th Edition
Authors: Steve Russo And Michael Silver
Question Posted:
Students also viewed these Sciences questions
-
GoPro has added the capability for recorded material to automatically sync between the cloud and the camcorder Which core marketing mix linkage does this represent Which supporting linkages could...
-
Planning is one of the most important management functions in any business. A front office managers first step in planning should involve determine the departments goals. Planning also includes...
-
KYC's stock price can go up by 15 percent every year, or down by 10 percent. Both outcomes are equally likely. The risk free rate is 5 percent, and the current stock price of KYC is 100. (a) Price a...
-
A horizontal jet of water (at 10C) that is 6 cm in diameter and has a velocity of 20 m/s is deflected by the vane as shown. If the vane is moving at a rate of 7 m/s in the x-direction, what...
-
Making a Decision as an Auditor: Effects of Errors on Income, Assets, and Liabilities Megan Company (not a corporation) was careless about its financial records during its first year of operations,...
-
A customer has approached a bank for a $100,000 one year loan at a 12% interest rate. If the bank does not approve this loan application, the $100,000 will be invested in bonds that earn a 6% annual...
-
What are the three parts of a make rule?
-
The Newburg Flyers operate a major sports franchise from a building in downtown Newburg. The building was built in 1940 at a cost of $5,000,000 and is fully depreciated so that it is shown on the...
-
5. Consider uniform, inviscid, supersonic flow exhausting from a rocket engine nozzle. For fixed back pressure p (i.e., the ambient pressure outside the nozzle), qualitatively draw the first few wave...
-
An acid is considered to be weak if: (a) It cannot neutralize a base. (b) It does not burn the skin. (c) Its equilibrium for dissociation lies far to the left. (d) It cannot produce hydronium (H 3 O...
-
Write a balanced dissociation equation for carbonic acid in water that shows the maximum number of H 3 O + ions the acid can yield.
-
A mortgage company offers to lend you $85,000; the loan calls for payments of $8,273.59 per year for 30 years. What interest rate is the mortgage company charging you?
-
After being stranded by his crew on an island in the Atlantic, Captain Kidd built a raft to escape and set out to sea on it. The wind is quite steady, at first blowing him due east for 19 km and then...
-
Farmer Joe is evaluating three (3) bids to supply fence hardware for the 5 acres of pasture that need to be fenced. Brady Fences Ltd. submits a bid of $45 per unit with a defect rate of 1.5%; Fence &...
-
Explain McGregor's Theories X and Y and how leaders apply them in the workplace
-
What is the energy conversion/transformation when you move a magnet near a coil and produce a current in the coil? Explain.
-
How does corporate strategy exert influence on capacity decisions within an organizational context?
-
How would you prioritize the considerations in menu planning for your restaurant?
-
You are the newly appointed tax practitioner to complete Emilys tax return and have downloaded the prefill report for Emilys tax return (hint, you can read what a prefill report is here (Links to an...
-
How many yellow lightwaves ( = 580 nm) will fit into a distance in space equal to the thickness of a piece of paper (0.003 in.)? How far will the same number of microwaves (v = 10 10 Hz, i.e., 10...
-
Establish that where A, α, b, and c are all constant, is a solution of the differential wave equation. This is a Gaussian or bell-shaped function. What is its speed and direction of...
-
Argon-ion lasers typically generate multi-watt beams in the green or blue regions of the visible spectrum. Determine the frequency of such a 514.5-nm beam.
-
Effective human resource management and organizational behavior as they relate to both Fair treatment and legal compliance
-
What would you describe to be one of your weaknesses? and strength?
-
Job fatigue and stress are significant problems faced by employees and their managers. Unfortunately, when a case of depression arises as a result, trying to resolve the problem may be...

Study smarter with the SolutionInn App


