These three substances are all liquids at room temperature: Which do you expect to be least soluble
Question:
These three substances are all liquids at room temperature:
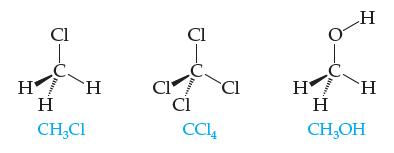
Which do you expect to be least soluble in water? Most soluble in water? Explain your answers fully
Transcribed Image Text:
Cl HEH Η H CH₂Cl CI Cl B Cl CC14 Cl H 1000 H H H CH₂OH
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
expect methanol CH3OH to be the most soluble in water and carbon tetrachloride CCl4 to be the least soluble in water Explanation Water is a polar mole...View the full answer

Answered By

GERALD KAMAU
non-plagiarism work, timely work and A++ work
4.40+
6+ Reviews
11+ Question Solved
Related Book For 

Introductory Chemistry Atoms First
ISBN: 9780321927118
5th Edition
Authors: Steve Russo And Michael Silver
Question Posted:
Students also viewed these Sciences questions
-
Planning is one of the most important management functions in any business. A front office managers first step in planning should involve determine the departments goals. Planning also includes...
-
You are analyzing various programs to reduce water pollution from food processing plants. In consultation with your staff, you have developed the following matrix of effects (where PV = Present...
-
Which do you expect to be more sustainable: grazing on public land or grazing on privately owned pastures? Why?
-
Capital Inc. has prepared the operating budget for the first quarter of 2015. They forecast sales of $50,000 in January, $60,000 in February, and $70,000 in March. Variable and fixed expenses are as...
-
JOURNAL ENTRIES FOR MATERIAL, LABOR, AND OVERHEAD Jones Manufacturing Corporation had the following transactions for its job order costing operation. Prepare general journal entries to record these...
-
Discuss the reasoning behind the assessment of classification models.
-
Using the variance data for Menounos Manufacturing Co., prepare an income statement through gross profit for the year ended December 31, 20Y6. Assume the company sold 3,000 units at $100 per unit.
-
Budget schedules for a manufacturer. Sierra Furniture is an elite desk manufacturer. It makes two products: Executive desks'3' ?? 5' oak desks Chairman desks'6' ?? 4' red oak desks The budgeted...
-
The current in the 1 3 . 8 2 resistor is / 3 = 0 . 9 A . 1 . Find the current in the other resistors in thecircuit? 2 . What is the total energy dissipated in resistor 4 1 7 . 2 0 if the power supply...
-
When a liquid solute dissolves in water, there is still a solute-separation step that absorbs energy, but the step doesnt require breaking up a crystal lattice as for a typical solid solute. What...
-
When a gaseous solute dissolves in water, which step in the dissolving process is essentially skipped? Explain why.
-
Use the method of cylindrical shells to find the volume generated by rotating the region bounded by the curves about the given axis. about the y-axis y = cos(Tx/2), y = 0, 0
-
Calculate B in terms of k, in the following equation of sums: n33 i=n7-33 7.9 05k6+4k7 +3k8+2k9 = k=n7
-
A cannonball is launched horizontally off a 50 m cliff with an initial velocity of 30 m/s. How far (in meters) is the cannonball from the base of the cliff when it hits the ground?
-
I've been reviewing our statements from the past year and need your help to verify a few entries. Last July, we fully processed orders of 760 units at a price of $38 resulting in $28,880 in revenue...
-
ABC is considering the purchase of a new computer system for the social marketing department. The system costs $375,000 and has an expected life of five years, salvage value of $15,000, and...
-
6. To cover some unexpected expenses, you take out a simple interest loan for $ 4500 at a rate of 6.1 % for up to one year. The lender allows you to repay the loan anytime during the year. a. Find...
-
In a medium-sized southern city, 86 houses are for sale, each having about 2000 square feet of floor space. The asking prices vary. The frequency distribution shown contains the price categories for...
-
Why is it necessary to study the diffusion of molecules in biological systems?
-
The steel shaft has a radius of 15 mm. Determine the torque T in the shaft if the two strain gages, attached to the surface of the shaft, report strains of ε x² = -80(10 -6 ) and...
-
The shaft has a radius of 15 mm and is made of L2 tool steel. Determine the strains in the x² and y² direction if a torque T = 2 kN · m is applied to the shaft. 45 VT
-
The A-36 steel pipe is subjected to the axial loading of 60 kN. Determine the change in volume of the material after the load is applied. 30 mm 40 mm 60 kIN 60 kN 0.5 m
-
Simplify 8. 219 9. (-2)6 10. (n) (n)(n+) 11. (4a) 12. (2mn)5 13. (5x) (7x) 14. -5(r-2) 15.-m(m+15) 16.9(b + 6b+8) 17. (5z - 1)z 18. (x+3)(x+9)
-
A person pushes on a stationary 125-N box with a force of 81 N at 30 below the horizontal, as shown in the figure. (Figure 1) The coefficient of static friction between the box and the horizontal...
-
a. (8 points) Calculate the potential difference V - Va due to the infinite rod of uniform charge density A, as shown below. Perform this calculation by directly integrating the electric field over a...

Study smarter with the SolutionInn App


