Using lines, solid wedges, and dashed wedges, draw the three-dimensional shape of chloromethylacetylene, C 3 H 3
Question:
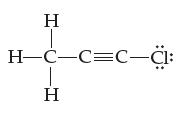
Using lines, solid wedges, and dashed wedges, draw the three-dimensional shape of chloromethylacetylene, C3H3Cl. Indicate the numeric value of all bond angles.
Transcribed Image Text:
HTCIH Η H-C-C=C-Cl: Η
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
Chemical structure of chloromethylacetylene C3H3Cl H HC CC CCl H Threedimensional shape of chloromet...View the full answer

Answered By

Charles mwangi
I am a postgraduate in chemistry (Industrial chemistry with management),with writing experience for more than 3 years.I have specialized in content development,questions,term papers and assignments.Majoring in chemistry,information science,management,human resource management,accounting,business law,marketing,psychology,excl expert ,education and engineering.I have tutored in other different platforms where my DNA includes three key aspects i.e,quality papers,timely and free from any academic malpractices.I frequently engage clients in each and every step to ensure quality service delivery.This is to ensure sustainability of the tutoring aspects as well as the credibility of the platform.
4.30+
2+ Reviews
10+ Question Solved
Related Book For 

Introductory Chemistry Atoms First
ISBN: 9780321927118
5th Edition
Authors: Steve Russo And Michael Silver
Question Posted:
Students also viewed these Sciences questions
-
Using lines, solid wedges, and dashed wedges, draw the three dimensional shape of ethane, C 2 H 6 . Indicate the numeric value of all bond angles. H=C=C- -
-
The connections in CH 3 NO 2 are: (a) Put in the remaining valence electrons to complete the Lewis dot diagram. (b) Using lines, solid wedges, and dashed wedges, draw the three-dimensional shape of...
-
The connections in C 2 N 2 H 2 O are: (a) Put in the remaining valence electrons to complete the Lewis dot diagram. (b) Using lines, solid wedges, and dashed wedges, draw the three-dimensional shape...
-
An individual can deduct for tax purposes the interest expense incurred on the mortgage loan attached to his or her personal residence. Is this statement true? Explain.
-
Ed Hickey, the consultant introduced at the beginning of section two of the chapter (p. 456), has obtained the following data relative to the Kaufman and D'Esti consulting job: Assume that the...
-
What is the difference between Chapter 7 and Chapter 11 bankruptcies? Why might Chapter 11 be better for claimants than Chapter 7?
-
1. Working in small groups, complete the Pay Raise Worksheet. 2. After the class reconvenes, group spokespersons present group findings. Pay Raise Worksheet April Knepper is the new supervisor of an...
-
Chrome-It, Inc., manufactures special chromed parts made to the order and specifications of the customer. It has two production departments, stamping and plating, and two service departments, power...
-
Beddington and May (1982) have proposed the following model to study the interactions between baleen whales and their main food source, krill (a small shrimp-like animal), in the southern ocean: - x...
-
Consider the molecules CH 4 , CH 3 Cl, CH 2 Cl 2 , CHCl 3 , and CCl 4 . Which are polar and which are nonpolar?
-
Using lines, solid wedges, and dashed wedges, draw the three-dimensional shape of the hydronium ion, H 3 O + . Indicate the numeric value of all bond angles. [H-Q-H]+ -C H
-
In r(t) is the position of a particle in space at time t. Find the angle between the velocity and acceleration vectors at time t = 0. - (Y21)i + (Y1 1612) j t 2 r(t) =
-
On 30 November 20x1, Systech Ltd entered into a non-cancellable contract to buy 1,000 shares of Fastrack Ltd for $5,000 on 31 July 20x2. On the same date, Systech Ltd purchased a put option on 1,000...
-
Explain the concept of a deductible temporary difference.
-
On 1 March 20x3, ABC Corporation, whose functional currency is the dollar, was informed that it had been successful in its tender for a contract to supply plant and equipment to an overseas customer....
-
Calculate consumption expenditure using the consumption function (as described by Equation 2) and the following estimates: Autonomous consumption: Autonomous consumption: $1,450 billion Income:...
-
After the press conference that followed the Federal Open Market Committee meeting on June 19, 2013, there were reports in the media that Chairman Bernankes comments were a signal that the Fed would...
-
This exercise should be used with Exercise 1-33B. Requirements 1. Prepare the income statement of Jill Carlson Banking Company for the year ended May 31, 2012. 2. What amount of dividends did Jill...
-
You have just begun your summer internship at Omni Instruments. The company supplies sterilized surgical instruments for physicians. To expand sales, Omni is considering paying a commission to its...
-
Identify the reagents you would use to convert 3-pentanone into 3-hexanone.
-
Identify the reagents necessary to achieve each of the following transformations. OEt OEt
-
Draw the structure of the product that is obtained when acetophenone is treated with each of following reagents: (a) Sodium hydroxide and excess iodine followed by H3O+ (b) Bromine in acetic acid (c)...
-
Recall from Chapter 1 that Dry Supply is a wholesaler of dry cleaning equipment, cleaning supplies, and laundry soap. The company, which is located in central Kansas, has been in business over 50...
-
This is an audit of Personal contributions> Concessional>Non concessional - What records would you expect to produce -? you have to name the records/documents etc
-
Figure 3 shows the velocity diagrams for 50% reaction and impulse turbines at different rotor speeds. The subscripts 2 and 3 denote conditions before and after the rotor respectively. In both cases,...

Study smarter with the SolutionInn App


