Using your knowledge of VSEPR theory and its preferred bond angles, suggest a reason for the fact
Question:
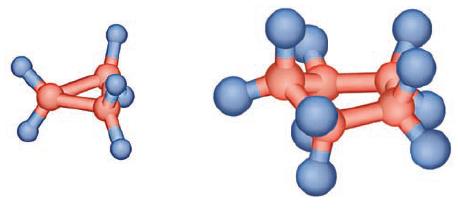
Using your knowledge of VSEPR theory and its preferred bond angles, suggest a reason for the fact that the cyclopropane molecule (pictured below) is not a very stable molecule and is highly reactive, while the cyclopentane molecule is not nearly as reactive or unstable.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
The cyclopropane molecule is not very stable and is highly reactive because of its ring strainRing strain is a type of energy that is caused by the de...View the full answer

Answered By

Keziah Thiga
I am a self motivated financial professional knowledgeable in; preparation of financial reports, reconciling and managing accounts, maintaining cash flows, budgets, among other financial reports. I possess strong analytical skills with high attention to detail and accuracy. I am able to act quickly and effectively when dealing with challenging situations. I have the ability to form positive relationships with colleagues and I believe that team work is great key to performance. I always deliver quality, detailed, original (0% plagirism), well-researched and critically analyzed papers.
4.90+
1504+ Reviews
2898+ Question Solved
Related Book For 

Introductory Chemistry Atoms First
ISBN: 9780321927118
5th Edition
Authors: Steve Russo And Michael Silver
Question Posted:
Students also viewed these Sciences questions
-
The explosion of an atomic bomb releases many radioactive isotopes, including strontium-90. Considering the location of strontium in the periodic table, suggest a reason for the fact that this...
-
The kinetic data for the radical chain chlorination of several cycloalkanes (see the adjoining table) illustrate that the C-H bonds of cyclopropane and, to a lesser extent, cyclobutane are somewhat...
-
The governance of an enterprise is the sum of those activities that make up the internal regulation of the business in compliance with the obligations placed on the firm by legislation, ownership and...
-
Not sure if this note is applicable to the problem? (A2) Prove there is a bijection between any two countably infinite sets.
-
South-Central Publishing Company prepares income statements segmented by divisions, but the chief operating officer is not certain about how the company is actually performing. Financial data for the...
-
The following data show the retail price for 12 randomly selected laptop computers along with their corresponding processor speeds in gigahertz. a. Develop a linear equation that can be used to...
-
The trial balance of Ninas Novelty, Inc., at September 30, 2010, and the data needed for the month-end adjustments follow: a. Insurance coverage still remaining at September 30, $800. b. Supplies...
-
Cole Petroleum has spent $206,000 to refine 63,000 gallons of petroleum distillate, which can be sold for $6.30 a gallon. Alternatively, Cole can process the distillate further and produce 53,000...
-
Buckingham and Clifton suggest ways to identify one's true talents. Apply their model to yourself. What do you find? Any surprises? What is authenticity? How do (and will) you find authenticity for...
-
What is it about the trigonal bipyramidal shape that distinguishes it from all the other molecular shapes that we have covered?
-
Cubane, C 8 H 8 , is an interesting molecule shown below. Comment on it relative to VSEPR. Then imagine you had dichloro cubane, C 8 H 6 C 12 . Draw all the unique molecules with this formula (you...
-
In Problem find f (x). f(x) = -7e x - 2x + 5
-
Why have the courts refused to interpret the Second Amendments protection of the right to keep and bear arms to prohibit gun control legislation? On what bases other than the Second Amendment can one...
-
What factors do you think a defense attorney considers in deciding whether to advise a client to testify in his or her own behalf at trial?
-
Should plea bargaining be abolished? If not, what modifications might be necessary to protect (a) the defendant and (b) the public?
-
Based on the ruling of the Supreme Court in City of Chicago v. Morales, what protections of the individual do you think must be included in an ordinance proscribing loitering?
-
How did the Jacksonville, Florida, vagrancy ordinance invalidated by the Supreme Court in the Papachristou case offend the Constitution of the United States? Have the reforms in vagrancy laws at the...
-
A survey conducted by CBS News asked 1,026 respondents: What would you do with an unexpected tax refund? The responses are summarized in the following table. ResponseFrequency Pay of...
-
A circular concrete shaft liner with Youngs modulus of 3.4 million psi, Poissons ratio of 0.25, unconfined compressive strength 3,500 psi and tensile strength 350 psi is loaded to the verge of...
-
Determine in each of the following cases if the function in the first column is an eigenfunction of the operator in the second column. If so, what is the eigenvalue? a. b. c. sine cos 3x (1/x) d/dx
-
If two operators act on a wave function as indicated by AÌBÌ f(x), it is important to carry out the operations in succession, with the first operation being that nearest to the...
-
Find the result of operating with d 2 /dx 2 + d 2 /dy 2 + d 2 /dz 2 on the function x 2 + y 2 + z 2 . Is this function an eigenfunction of the operator?
-
Assume that all reactants and products are in their standard states and use reduction potential data from (Petrucci's Appendix D; OpenStax's Table 16.1) to predict whether a spontaneous reaction will...
-
Use the Nernst equation and reduction potential data from (Petrucci's Appendix D; OpenStax's Table 16.1) to calculate Ecell for the following cell: Al (s) | Al3+ (0.18 M) || Fe+ (0.85 M) | Fe (s)
-
Osmosis is the net movement of water across a semipermeable membrane from an area of lower solute concentration to an area of higher solute concentration. The water will continue to move across the...

Study smarter with the SolutionInn App


