Which hydrocarbon molecule is least unsaturated? Explain your answer. (a) = (b) (c)
Question:
Which hydrocarbon molecule is least unsaturated? Explain your answer.
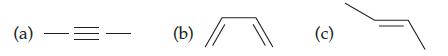
Transcribed Image Text:
(a) —= (b) (c)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
The molecule in the image c is the least unsaturated It has the form...View the full answer

Answered By

Milan Mondal
I am milan mondal have done my Msc in physics (special astrophysics and relativity) from the University of burdwan and Bed in physical science from the same University.
From 2018 I am working as pgt physics teacher in kendriya vidyalaya no2 kharagpur ,west bengal. And also I am doing advanced physics expert in chegg.com .also I teach Bsc physics .
I love to teach physics and acience.
If you give me a chance I will give my best to you.
5.00+
4+ Reviews
10+ Question Solved
Related Book For 

Introductory Chemistry Atoms First
ISBN: 9780321927118
5th Edition
Authors: Steve Russo And Michael Silver
Question Posted:
Students also viewed these Sciences questions
-
Draw all possible line drawings for the unsaturated hydrocarbon molecule in Practice Problem 17.5. Data from Practice Problem 17.5 A particular linear hydrocarbon molecule has six carbons and ten...
-
The structure of a lipid bilayer is determined by the particular properties of its lipid molecules. What would happen if: A. Phospholipids had only one hydrocarbon tail instead of two? B. The...
-
A particular linear hydrocarbon molecule has six carbons and ten hydrogens. Is it unsaturated or saturated?
-
Explain the Mechanism & routes of administration of SEDDS (Self emulsifying drug delivery system).
-
Seventy-two percent of the members of the United Food and Commercial Workers Local 655 voted to strike against Stop n Shop in the St. Louis area. In fear of similar union responses, two of Stop n...
-
Phoenix Tool Company and Denver Tool Company have had a very similar record of earnings performance over the past eight years. Both firms are in the same industry and, in fact, compete directly with...
-
On August 6, it was 80o F in New York City, so it was twice as hot as on December 7, when it was 40 F. Determine whether the given statement represents a meaningful ratio, so that the ratio level of...
-
Assume Nordican Boot Company is considering making a specially designed snow boot just for the Winter Olympics. Which of the following costs or activities would probably be differential in...
-
Caesar's information system can be used to access whether the placement of slot machines affects profitability. Who would use this information?
-
The difference between an alkane and an alkene is: (a) Neither has a triple bond between carbon atoms. (b) Alkanes are unsaturated whereas alkenes are saturated. (c) Alkanes have at least one double...
-
Draw a dot diagram for methanoic acid, showing all hydrogen, carbon, and oxygen atoms.
-
Suppose that signs of an improvement in the Japanese economy lead international investors to resume lending to the Japanese government and busi- nesses. How would this event affect the market for the...
-
The FDIC is extremely concerned with risk management in banks. High-risk banks are more likely to fail and cost the FDIC money. The FDIC regularly examines banks and rates them using a system called...
-
The Securities and Exchange Commission is responsible for regulating securities firms. Go to www.sec.gov. This is the official home page of the SEC. Use this page to answer the following. a. What is...
-
Consider an economy characterized by the following facts: i. The official budget deficit is \(4 \%\) of GDP. ii. The debt-to-GDP ratio is \(100 \%\). iii. The nominal interest rate is \(10 \%\). iv....
-
The following site can be used to demonstrate how the features of an option affect the option's prices. Go to http://www.hoadley.net/options/bs.htm. Scroll down to the online options calculator. What...
-
In 2004, Congress allocated over $20 billion to fight illegal drugs. About 60 percent of the funds was directed at reducing the supply of drugs through domestic law enforcement and interdiction. Some...
-
Chopra Company, which makes golf carts and lawn tractors, uses departmental rates to allocate overhead costs. It uses direct materials costs as the basis for allocating overhead in Department A....
-
The Higher the time period of the financial security the higher the. ............... risk. O a. Maturity O b. Default and Maturity Oc. Default O d. Liquidity
-
Dipoles A and B are both located in the field of a point charge Q, as shown in Fig. 20.27. Does either experience a net torque? A net force? If each dipole is released from rest, describe...
-
Can electric field lines ever cross? Why or why not?
-
The electric flux through a closed surface is zero. Must the electric field be zero on that surface? If not, give an example.
-
1. Given the network diagram (assume finish-to-start relationships and duration in days): ES ID EF TFFF LS DUR LF OAS // 88 1000 SC36 01/10 8 28 36 36 E 80 8 11/8 44 44 88 36 F88 36 52 88 88 H114 88...
-
The simple mechanism shown in the figure can be calibrated to measure temperature change. Use dimensions of a = 26 mm, b = 88 mm, and L = 166 mm. The coefficient of thermal expansion for member (1)...
-
A solid cylinder of diameter d = 80 mm is fixed at A. A rigid bar is welded at the other end as shown. A force F is applied in the z-direction and a force 10F is applied in the x-direction. For the...

Study smarter with the SolutionInn App


