Which of the following are electron-transfer reactions? For those that are, indicate which reactant is the reducing
Question:
Which of the following are electron-transfer reactions? For those that are, indicate which reactant is the reducing agent and which reactant is the oxidizing agent.
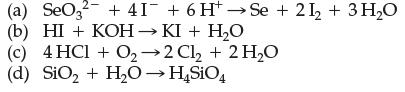
Transcribed Image Text:
(a) SeO3² +41 + 6 H→Se +21₂ + 3H₂O (b) HI + KOH → KI + H₂O (c) 4 HCl + O2-2 Cl₂ + 2 H₂O (d) SiO₂ + H₂O → H₂SiO4
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
To determine which of the given reactions are electrontransfer reactions and identify the reducing a...View the full answer

Answered By

Vijesh J
My passion to become a tutor is a lifetime milestone. Being a finance and marketing professional with hands-on experience in wealth management, portfolio management, team handling and actively contributing in promoting the company. Highly talented in managing and educating students in most attractive ways were students get involved. I will always give perfection to my works. Time is the most important for the works and I provide every answer on time without a delay. I will proofread each and every work and will deliver a with more perfection.
4.70+
5+ Reviews
15+ Question Solved
Related Book For 

Introductory Chemistry Atoms First
ISBN: 9780321927118
5th Edition
Authors: Steve Russo And Michael Silver
Question Posted:
Students also viewed these Sciences questions
-
For those reactions in Problem 10.58 that are electron-transfer reactions: (a) Indicate which atoms get oxidized and which atoms get reduced. (b) Indicate which reactant is the oxidizing agent. (c)...
-
For those reactions in Problem 10.56 that are redox reactions: (a) Indicate which atoms get oxidized and which atoms get reduced. (b) Indicate which reactant is the oxidizing agent. (c) Indicate...
-
Which of the following are electron-transfer reactions? For those that are, indicate which reactant is the reducing agent and which reactant is the oxidizing agent. (a) PF 3 + 3H 2 O H 3 PO 3 + 3HF...
-
Question)please make sure also calculate Total and Annual MacDUR. Find the Macaulay Duration for a \( 12 \% \) semiannual coupon bond with \( \$ 1,000 \) face, 3 years to maturity, and a YTM of \(...
-
A corporation issued $6,000,000 of 20-year bonds for cash at 104. How would the transaction be reported on the statement of cash flows?
-
The cash account for Freds Sports Co. on June 1, 2010, indicated a balance of $16,515. During June, the total cash deposited was $40,150, and checks written totaled $43,600. The bank statement...
-
A burst of compressed air pushes a pellet out of a blowpipe. The force exerted by the air on the pellet is given by \(F(t)=F_{0} e^{-t / \tau}\), where \(\tau\) is called a time constant because it...
-
Burtonville Stores accepts both its own and national credit cards. During the year, the following selected summary transactions occurred. Jan. 15 Made Burtonville credit card sales totaling $18,000....
-
An investment pays $500 at the end of each month for four years and then a lump sum payment of $150,000 at the end of four years. The quoted annual discount rate is 6%. What is the PV of this...
-
Assign an oxidation state for each nitrogen atom in N 3 .
-
A battery was produced using copper metal in a solution of Cu 2+ ions connected to rhodium metal in a solution of Rh 3+ ions. Copper is the anode and rhodium is the cathode. Is rhodium higher or...
-
Find the frequency domain voltage V0 as shown. Ao06-701
-
Do the ISO standards require that a "goal" for an LCA be clearly stated?
-
Tax costs may be avoided in its entirety when the transaction is structured as what type of reorganization?
-
Why is EEG equipment inexpensive and portable compared to other neuroimaging techniques?
-
What activity would have triggered red flags? Describe the transactional activity.
-
Why would a company want to use account numbers on its Chart of Accounts?
-
Explain why each of the following payments does or does not meet the IRS's definition of a tax: a. Jack is a licensed beautician. He pays the state $45 each year to renew his license to practice as a...
-
What steps must a business take to implement a program of social responsibility?
-
For each of the following sets of operating conditions, list at least one appropriate type of pump. See Fig. 13.52. a. 500 gal/min of water at 80 ft of total head b. 500 gal/min of water at 800 ft of...
-
Describe the effect of operating two pumps in series.
-
If two identical centrifugal pumps are connected in parallel and operated against a certain head, how would the total capacity compare with that of a single pump operating against the same head?
-
A pension fund manager is considering three mutual funds. The first is a stock fund, the second is a long-term government and corporate bond fund, and the third is a T-bill money market fund that...
-
A company named ABC hired three employees in different years; each employee gets an increment of 5000 yearly. Also each employee gets bonus amount of 10k for each project he has done yearly. The...
-
You are scheduled to receive $9000 in 3 years. When you receive it, you will invest it for 12 years at a rate of 5.50%. How much will you have at the end of 15 years? Present value Discount rate (r)...

Study smarter with the SolutionInn App


