Which of the following are substitution reactions: (a) CH3Br + ICHI + Br (b) H + Br2
Question:
Which of the following are substitution reactions:
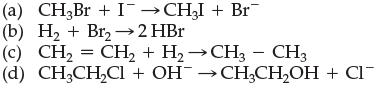
Transcribed Image Text:
(a) CH3Br + I¯→CH₂I + Br (b) H₂ + Br2 → 2 HBr (c) (d) - CH, = CH, + H,→CH → CH3 CH3CH₂Cl + OH¯ →CH₂CH₂OH + CI
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
Explanation a This is a substitution reaction because bromine is replaced by iodine from met...View the full answer

Answered By

Aqib Parvej
I am teaching since my graduation time so I have teaching experience of about 5 years and in these years I learn to teach in the best and interesting way .
4.80+
20+ Reviews
41+ Question Solved
Related Book For 

Introductory Chemistry Atoms First
ISBN: 9780321927118
5th Edition
Authors: Steve Russo And Michael Silver
Question Posted:
Students also viewed these Sciences questions
-
Suppose the output of a factory (output) and the number of workers (workers) is represented by a linear function output= a + b workers, where a and b are constants. You know that when there are 5...
-
Healthtech Foods managers are considering developing a new low-fat snack food. It is to be a blend of two types of cereals, each of which has different fiber, fat, and protein characteristics. The...
-
Which of the following alkyl halides form a substitution product in an SN1 reaction that is different from the substitution product formed in an SN2 reaction? a. b. c. d. e. f. CH Br CH CHCHCHCH CHa...
-
Watch the sample Persuasive Presentation Video link given in this module on Success is a Continuous Journey by Richard St. John .Answer the following questions in detail and use complete sentences....
-
Define accounting.
-
Hook Industries capital structure consists solely of debt and common equity. It can issue debt at rd = 11%, and its common stock currently pays a $2.00 dividend per share (D0 = $2.00). The stocks...
-
A hydrogen atom is excited from its ground state to the \(n=\) 4 state. The atom subsequently emits three photons, one of which has a wavelength of \(656 \mathrm{~nm}\). What are the wavelengths of...
-
Your employer, a mid-sized human resources management company, is considering expansion into related fields, including the acquisition of Temp Force Company, an employ mentagency that supplies word...
-
How does MariaDB leverage optimizer enhancements such as Cost-Based Optimization (CBO), Index Condition Pushdown (ICP), and Histogram-Based Statistics to improve query execution efficiency and plan...
-
The reaction N 2 + 3H 2 2 NH 3 is exothermic. Draw a reaction-energy profile for the reaction. Label the gap that represents E rxn .
-
E rxn for the reaction XSY is +30 kJ. (a) Is the reaction endothermic or exothermic? (b) Rewrite the reaction showing heat as either a reactant or a product. (c) What is the value of E rxn for the...
-
What is the vested benefit obligation?
-
Based on the assessment of Rent The Runway's business in January 2010, if you were Hyman or Fleiss, would you proceed with scaling (accelerated business growth) at this time? What are the...
-
Provide an example of an effective manager and leader in a criminal justice organization. Include common characteristics IN CRIMINAL JUSTICE.
-
Managing unplanned disruptions through business continuity planning requires organizations to develop Enterprise Resilience, briefly explain the relationship between Business Continuity and...
-
Joanne Smith was born in 1948. John Smith, Joanne's husband, was born in 1940. They are both not blind. They gave these following people more than 50% of their support throughout the year. Their...
-
Drawing on relevant business continuity and or crisis management framework, can you discuss the relationship between crisis management, business continuity, and risk management and critically discuss...
-
Ozzello Property Management is organized as a partnership. The owners, Lorenzo, Erwin, and Michelle, share profits and losses 30:30:40. Ozzello has the following results for the current year:...
-
The registrar of a college with a population of N = 4,000 full-time students is asked by the president to conduct a survey to measure satisfaction with the quality of life on campus. The following...
-
Your friends car has broken down, and you volunteer to push it to the nearest repair shop, which is 2.0 km away. You carefully move your car so that the bumpers of the two cars are in contact and...
-
A tall strongman of mass m = 95 kg stands upon a scale while at the same time pushing on the ceiling in a small room. Draw a free-body diagram of the strongman (Fig. P3.34) and indicate all normal...
-
A bodybuilder configures a leg-press apparatus (Fig. P3.35) to a resistance of 1000 N (about 230 lb). She pushes the weight to her full extension and comes to rest. (a) What is the normal force on...
-
The last step of the change management process that we used in previous weeks is to develop a pilot test to begin to make real change. Since the problems we are addressing are complex, it is not so...
-
Solve each of the following recurrence relations by expanding it out and solving the resulting summation. Your answer should be a function in terms of N. (a) T(N) T(N-1) - 10; T(1)=5 (b) T(N)...
-
The team facilitator is discussing with key stakeholders the status of the project and showing them the tasks that are remaining, the tasks that are currently being worked on, and the ones completed....

Study smarter with the SolutionInn App


